INTRODUCTION
Stress is the physiological responses of organisms to harmful or threatening stimuli that allow appropriate behavioral responses to the stressor. If the organism could not adapt to stress, adaptive physiological responses are converted into the maladaptive pathological conditions (Borodovitsyna et al., 2018). Several interconnected brain areas are involved in the control of stress and anxiety. Especially, locus coeuleus, amygdala, and hypothalamic-pituitary-adrenal (HPA) system are the key components in the control of stress and anxiety (Adhikari, 2014). The locus coeuleus is the major central noradrenergic nucleus and coordinates behavioral and autonomic responses related to anxiety, fear, and stress (Berridge and Waterhouse, 2003). The hypothalamic paraventricular nucleus regulates activity of the HPA axis and connects the brain to the neuroendocrine stress response system (Herman and Tasker, 2016). Dysregulation of these brain areas increases the risk for anxiety- and stress-related disorders (Myers et al., 2017).
The transcription factor c-Fos is rapidly induced in response to cellular activity, and c-Fos regulates expression of multiple target genes. c-Fos is increased in various brain regions of mice and rats exposed to chronic stress, especially in the paraventricular nucleus of the hypothalamus (Imaki et al., 2003). Therefore, induction of c-Fos has been used as an indicator of neuronal activation in the key areas, paraventricular nucleus and locus coeuleus. c-Fos is specifically linked to neuronal plasticity, long-term potentiation, and synapse formation, and alterations in synaptic connectivity could be an underlying mechanism that leads to long-term changes in emotionality following acute stress exposure (Minatohara et al., 2016; Ploski et al., 2011).
Nitric oxide synthase (NOS) regulates regional neuronal activation depending upon the stimulus (Sherwin et al., 2017). Nitric oxide (NO) is synthesized by a number of NOS isoenzymes, which catalyze the oxidation of L-arginine to NO and citrulline. NOS is implicated in the anxiety-associated behaviors (Miguel and Nunes-de-Souza, 2008; Workman et al., 2008). There are three isoforms of NOS: neuronal NOS (nNOS), endothelial NOS and inducible NOS. nNOS overexpression in the hippocampus is essential for chronic stress-induced depression and inhibition of nNOS signaling in the brain may represent a novel approach for the treatment of depressive disorders (Zhou et al., 2007).
Regular exercise improves mental health for individuals with anxiety disorders (Asmundson et al., 2013; Hovland et al., 2013; Merom et al., 2008). However, a little information is available regarding the effect of treadmill exercise on the stress-induced anxiety disorder. In the present study, the effect of treadmill exercise on stress-induced anxiety focusing on the expressions of c-Fos and nNOS was investigated using rats.
MATERIALS AND METHODS
Experimental animals and treatments
The experimental procedures were performed in accordance with the animal care guidelines of the National Institutes of Health and the Korean Academy of Medical Sciences. Male Sprague-Dawley rats, weighing 100±5 g (5 weeks old), were used in this experiment (Orient Co., Seoul, Korea). Each animal was housed under controlled temperature (20°C±2°C) and lighting (07:00 a.m. to 19:00 p.m.) conditions with food and water made available ad libitum. The animals were randomly divided into 4 groups (n=9 in each group): the control group, the exercise group, the stress-induced group, and the stress-induced and exercise group.
Induction of stress
Stress was induced by applying inescapable 0.2 mA electric foot shock to the rats, as the previously described method (Kim et al., 2015). One set of electric foot shock composed of 6-sec duration and repeated 10 times with 30-sec interval. The rats in the stress-induced groups received three sets of electric foot shock per day. Exposure of rats to the electric foot shock continued for 7 days.
Exercise protocol
The rats in the exercise groups were forced to run on a motorized treadmill for 30 min once a day for 4 weeks, stating 1 day after last electric food shock. The exercise load consisted of running at a speed of 5 m/min for the first 5 min, 8 m/min for the next 5 min, and 15 m/min for the last 20 min, with a 0° inclination.
Open field test
For the locomotor activity, open field test was conducted, as the previous described method (Kim et al., 2011). The animals were randomly assigned to an order of testing and placed in a white square open field arena (100×100 cm) made of wood. It was enclosed with 40-cm-high walls and placed under strong illumination (200 lux). The arena was divided into 25 squares (each square is 20×20 cm), defined as 9 central and 16 peripheral squares. The animal was placed in the center of the arena and left free to explore the environment for 1 min. After that time, the number of squares that the rat crossed was recorded for 5 min.
Elevated plus-maze test
Anxiety-like behaviors were evaluated using the elevated plus-maze test, as the previous described method (Kung et al., 2010; Sterley et al., 2011). The elevated plus-maze test consisted of two opposing open arms (45×10 cm) and two closed arms (45×10× 50 cm) that extended from a central platform (10×10 cm) elevated 65 cm above the floor. Each rat was placed on the central platform facing a closed arm, and was allowed to freely explore the maze for 5 min. Entry into an arm was defined as entry of all four paws into the arm. The time spent in the open arms and the percentage of the entries in the open arms during 5 min was calculated.
Tissue preparation
The animals were sacrificed immediately after finishing open field test. The animals were anesthetized using ethyl ether after which the rats were transcardially perfused with 50 mM phosphate-buffered saline (PBS), and fixed with a freshly prepared solution consisting of 4% paraformaldehyde in 100 mM phosphate buffer (pH, 7.4). The brains were dissected and post-fixed in the same fixative overnight and transferred into a 30% sucrose solution for cryoprotection. Coronal sections of 40 μm thickness were made with a freezing microtome (Leica, Nussloch, Germany).
Immunohistochemistry for c-Fos and nNOS
Immunohistochemistry for c-Fos-positive and nNOS-positive cells in the hypothalamus (Bregma −1.80 to −1.92 mm) and locus coeruleus (Bregma −9.84 to −9.96 mm) was conducted, as the previous described method (Seo et al., 2013; Sim et al., 2008). The sections were incubated in PBS for 10 min, and then washed three times in the same buffer. The sections were then incubated in 1% H2O2 for 30 min. Next, the sections were incubated overnight with rabbit anti-c-Fos antibody (1:500; Santa Cruz Biotechnology, Santa Cruz, CA, USA) or mouse anti-nNOS antibody (1:500; BD Biosciences, San Jose, CA, USA). The sections were then incubated for 1 hr with anti-rabbit secondary antibody (1:200; Vector Laboratories, Burlingame, CA, USA) for c-Fos immunohistochemistry and with anti-mouse secondary antibody (1:200; Vector Laboratories) for nNOS immunohistochemistry. Next, the sections were incubated with avidin-biotin-peroxidase complex (1:100; Vector Laboratories) for 1 hr at room temperature. For staining, the sections were incubated in solution consisting of 0.02% DAB and 0.03% H2O2 in 50 mM Tris-HCl (pH, 7.6) for approximately 5 min, after which they were washed with PBS and mounted onto gelatin-coated slides. The numbers of c-Fos and nNOS-positive cells in the hypothalamus and locus coeruleus were counted using a light microscope (Olympus, Tokyo, Japan).
RESULTS
Anxiety-like behaviors
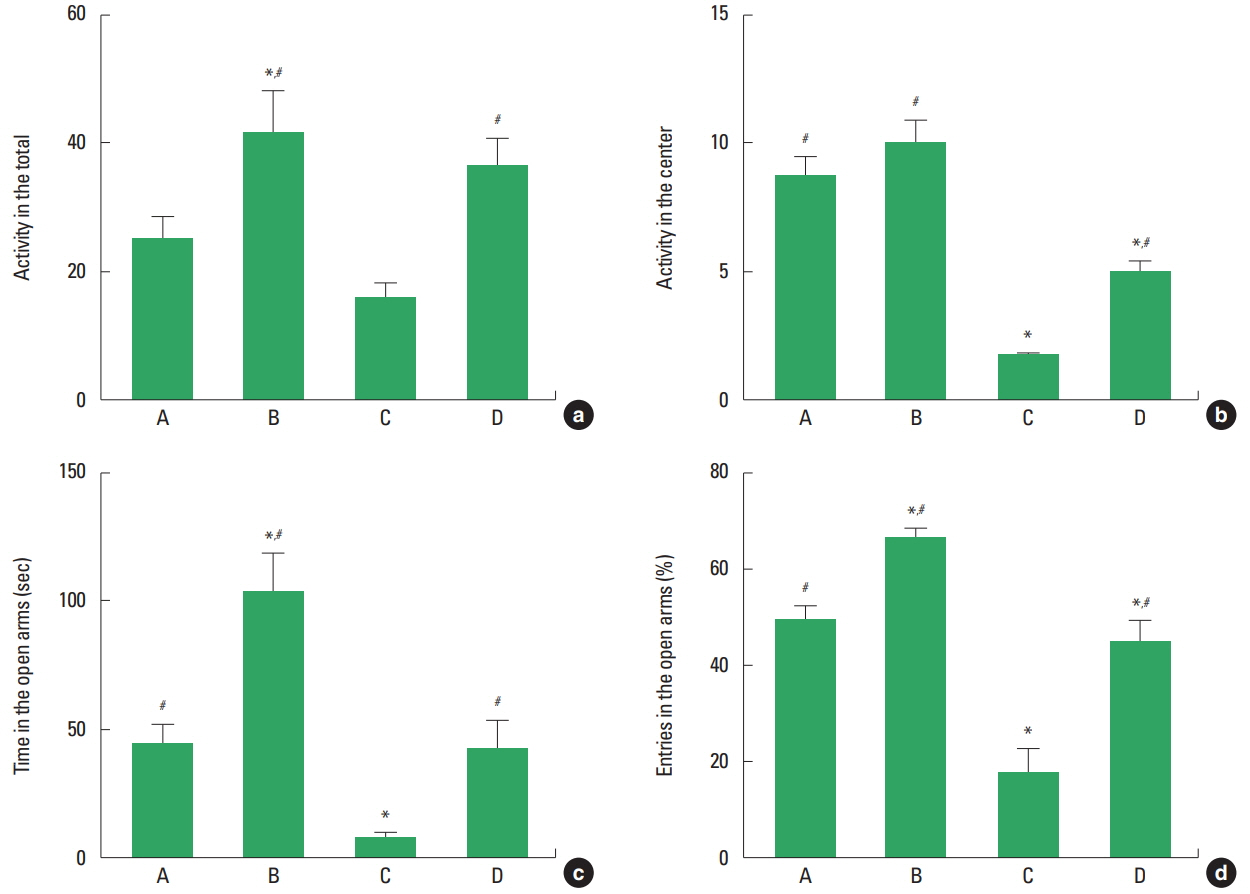
The effect of treadmill exercise on locomotor activity is presented in Fig. 1a, b. Activity in the inner zone was decreased in the stress-induced group compared to the control group (P<0.05). Treadmill exercise increased activity both in the control group and in the stress-induced group (P<0.05).
The effect of treadmill exercise on anxiety-like behaviors is presented in Fig. 1c, d. The time spent in the open arms and the percentage of the entries in the open arms was lower in the stress- induced group compared to the control group (P<0.05). Treadmill exercise increased time spent and the percentage of the entries both in the control group and in the stress-induced group (P<0.05).
Expressions of c-Fos and nNOS in the hypothalamus
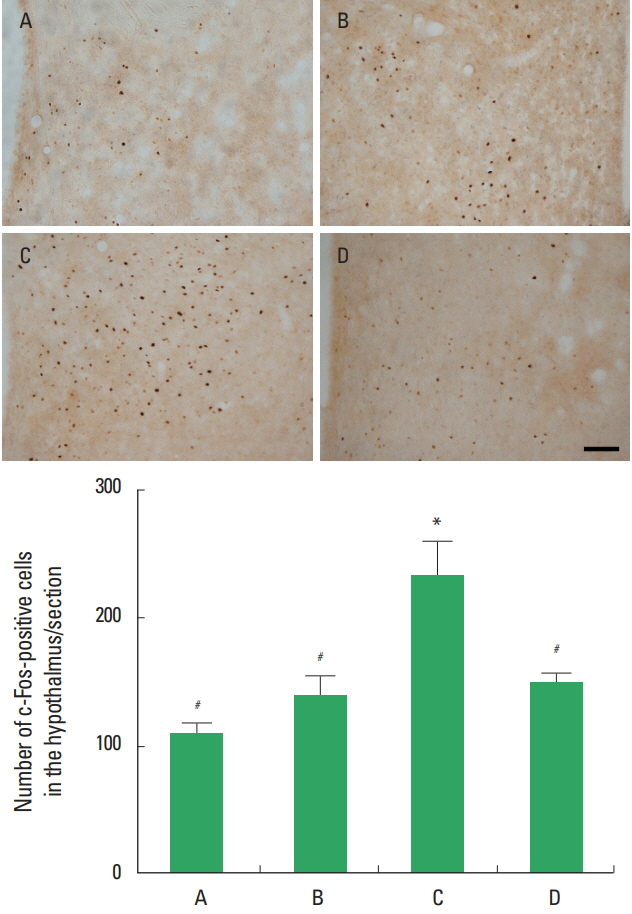
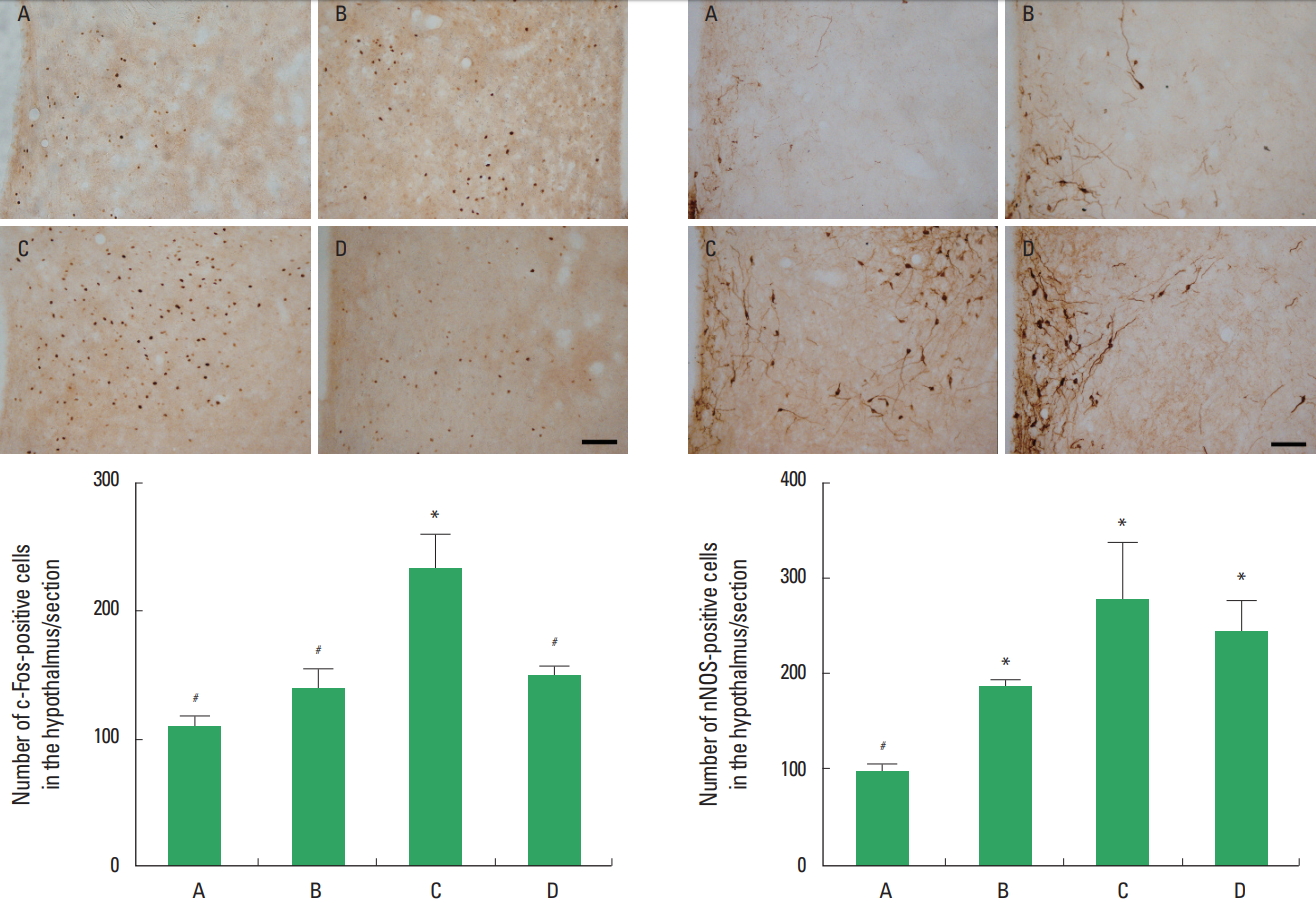
The number of c-Fos-positive cells in the hypothalamus was presented in Fig. 2. c-Fos expression in the hypothalamus was increased in the stress induced group compared to the control group (P<0.05). Treadmill exercise suppressed c-Fos expression in the hypothalamus of the stress induced group (P<0.05).
The number of nNOS positive cells in the hypothalamus was presented in Fig. 3. nNOS expression in the hypothalamus was increased in the stress induced group compared to the control group (P<0.05). Treadmill exercise exerted no significant effect on nNOS expression in the hypothalamus of the stress induced group (P>0.05).
Expressions of c-Fos and nNOS in the locus coeruleus
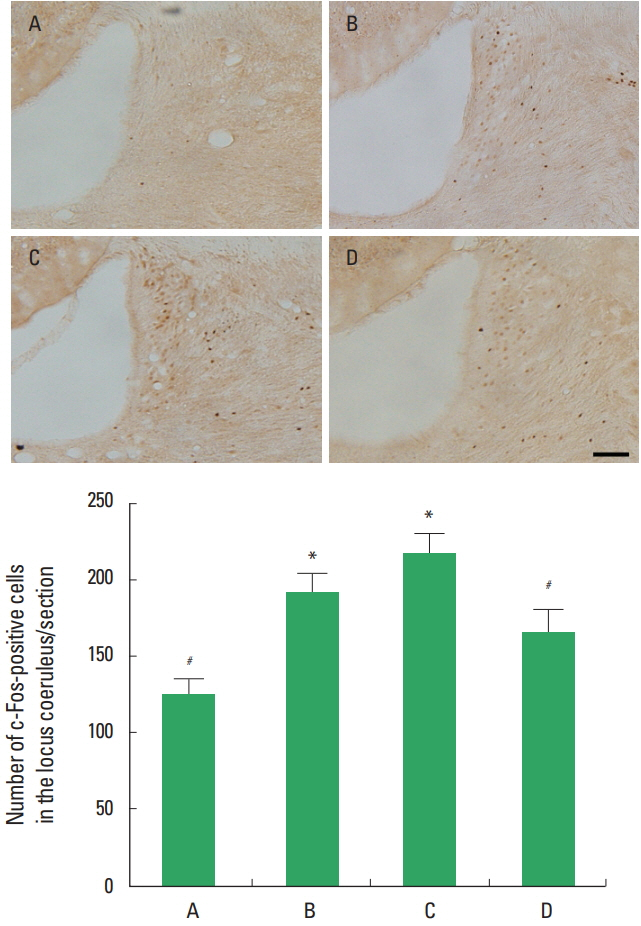
The number of c-Fos-positive cells in the locus coeruleus was presented in Fig. 4. c-Fos expression in the locus coeruleus was increased in the stress induced group compared to the control group (P<0.05). Treadmill exercise suppressed c-Fos expression in the locus coeruleus of the stress induced group (P<0.05).
The number of nNOS positive cells in the locus coeruleus was presented in Fig. 5. nNOS expression in the locus coeruleus was increased in the stress induced group compared to the control group (P<0.05). Treadmill exercise suppressed c-Fos expression in the locus coeruleus of the stress induced group (P<0.05).
DISCUSSION
Newman and Matta (2007) reported that aerobic exercise reduced childhood posttraumatic stress disorder (PTSD), depression, and anxiety. Anxiolytic effect of exercise is well documented (Asmundson et al., 2013; Merom et al., 2008). The distance traveled in the open field test is a common measure of exploratory and spontaneous locomotor activity in rodents (Verma et al., 2010; Zhao et al., 2016). The rats with anxiety spent a lot of time in the open field test (Seibenhener and Wooten, 2015). The elevated plus-maze reflects passive avoidance of an animal in response to a potential threat and has become a very popular approach for the evaluation of anxiety behaviors by assessing the number of open arm entries as well as the time spent in open arms (Lister, 1987). Long-term treadmill running in rats reduced anxiety-like behaviors in the elevated plus-maze test (Fulk et al., 2004).
In the present study, locomotor activity in the center of the open field test and the number of entries and time in the open arms of the elevated plus-maze test were reduced in the rats with stress-induced anxiety. Treadmill running enhanced the locomotor activity with the number of entries and time in the stress-induced anxiety rats.
Expression of c-Fos following stress is highly region-specific, including the paraventricular nucleus and locus coeruleus, and c-Fos expression is involved in the adaptation to social stress (Martinez et al., 1998). c-Fos expression in the paraventricular nucleus and locus coeruleus was increased by single restraint, and this increment was attenuated by repeated restraint stress (Kwon et al., 2006). c-Fos expression represents neuronal activity and plays a crucial role in the brain development (Sim et al., 2008). Restraint stress enhances c-Fos expression in the paraventricular nucleus, locus coeruleus, supraoptic nucleus, rostral raphe pallidus, nucleus of the solitary tract and ventrolateral medulla (Goebel et al., 2009). Treadmill exercise decreased the expression of c-Fos in the PTSD-induced rats, indicating that treadmill exercise during pregnancy suppressed neuronal activation induced by the stress exposure (Seo et al., 2013).
In the present study, c-Fos expression in the hypothalamus and locus coeruleus was increased in the stress-induced rats. Treadmill exercise reduced c-Fos overexpression in the stress-induced rats. The present results demonstrated that treadmill exercise alleviated the stress-induced c-Fos expression.
Inhibition of nNOS altered social interactions and decreased anxiety-like behaviors (Forestiero et al., 2006). Inhibition of nNOS signaling in the brain may be a novel approach for the treatment of depressive disorders (Zhou et al., 2007). Inhibition of NOS caused antidepressive effect (Jesse et al., 2008; Seo et al., 2013).
In the present study, nNOS expression in the hypothalamus and locus coeruleus was increased in the stress-induced rats. Treadmill exercise reduced nNOS overexpression in the stress-induced rats. The present results demonstrated that treadmill exercise alleviated the stress-induced nNOS expression.
The present results showed that treadmill exercise ameliorated anxiety-like behaviors in the stress-induced rats. This effect of treadmill exercise on anxiety-like behaviors might be ascribed to the suppressing effect of exercise on c-Fos and nNOS expressions.