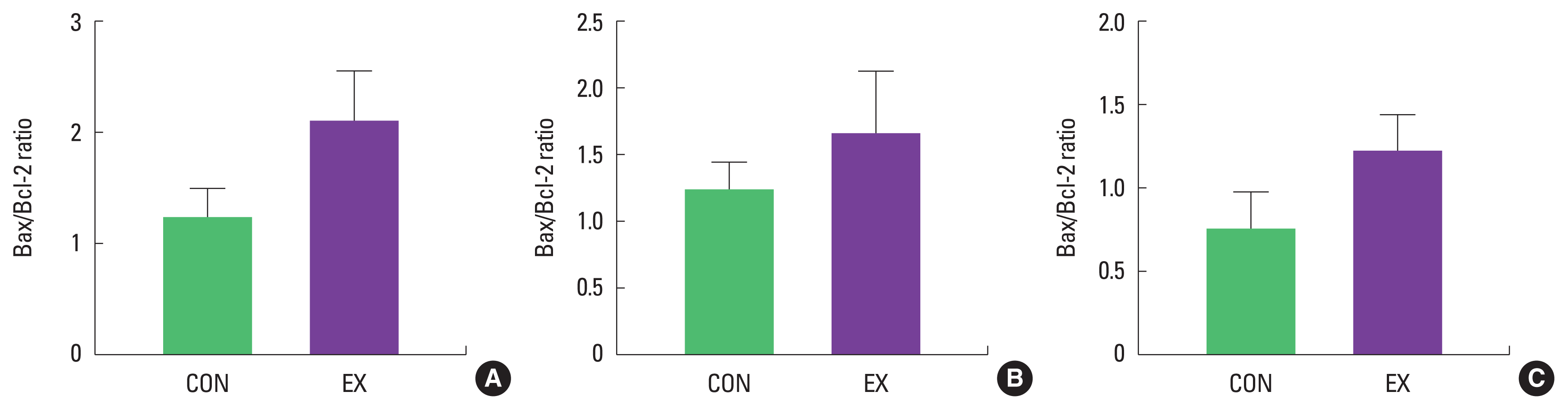Effects of a single bout of exercise on mitochondria-mediated apoptotic signaling in rat cardiac and skeletal muscles
Article information
Abstract
This study aimed to determine the effects of a single bout exercise on mitochondria-mediated apoptotic signaling in cardiac and skeletal muscles. Fischer 344 rats (4 months old) were randomly divided into the control or a single bout of exercise group (n=10 each). The rats performed a single bout of treadmill exercise for 60 min. Mitochondria-mediated apoptotic signaling (e.g., Bax, Bcl-2, mitochondrial permeability transition pore [mPTP] opening, cytochrome c, and cleaved caspase-3) was measured in cardiac (e.g., left ventricle) and skeletal (e.g., soleus and white gastrocnemius) muscles. A single bout of exercise significantly decreased mPTP opening sensitivity in all tissues. However, a single bout of exercise did not show any statistical differences in Bax, Bcl-2, cytochrome c, and cleaved caspase-3 in all tissues measured. A single bout of exercise did not show definite results on characteristics of mitochondria-mediated apoptotic signaling. Therefore, further research is necessary to provide a more mechanistic understanding of the apoptosis pathway.
INTRODUCTION
Mitochondria are the powerhouses responsible for the energy production that occurs in cells called adenosine triphosphate (ATP) synthesis (Murphy et al., 2016). In addition to providing cellular energy, mitochondria are involved in various tasks, such as cellular respiration, oxidative stress, and calcium homeostasis. Mitochondria also conduct mitochondrial quality control; (a) maintaining mitochondrial morphology, integrity, number, and size through cycles of mitochondrial dynamics (fusion and fission); (b) it promotes the turnover of damaged mitochondria and inhibits the accumulation of dysfunctional mitochondria through mitochondrial autophagy (mitophagy) (Yoo et al., 2019).
Mitochondria can serve as mediators, coordinating a highly programmed cell death referred to as apoptosis (Redza-Dutordoir and Averill-Bates, 2016). According to previous studies, dysfunctional mitochondria and mitochondrial reactive oxygen species (mtROS) induce the initial events of mitochondria-mediated apoptosis by releasing pro-apoptotic proteins into the cytoplasm (Redza-Dutordoir and Averill-Bates, 2016; Xiong et al., 2014). Imbalance between pro-apoptotic proteins (e.g., Bax and Bid) and antiapoptotic proteins (e.g., Bcl-2 and Bcl-xl) induces the opening of the mitochondrial permeability transition pore (mPTP) and the release of cytochrome c which serves as an electron transporter in the electron transfer chain of inner mitochondrial membranes. The mitochondria-released cytochrome c binds to ATP, apoptotic protease-activating factor 1, and pro-caspase 9, which activates caspase-3, consequently resulting in DNA fragmentation (Heo et al., 2017; Xiong et al., 2014).
It is well known that exercise is vital for health and fitness because it improves immune function, cardiovascular system function, and metabolic function (Gleeson et al., 2011). From a cellular and molecular point of view, chronic exercise training not only promotes mitochondrial function (Gram et al., 2015) and mitochondrial quality control (Konopka et al., 2014; Tarpey et al., 2017) but it also impedes apoptosis (Peterson et al., 2008). For these reasons, studies on the effects of chronic exercise have been mainly carried out at the cellular and molecular level in cardiac and skeletal muscle (Gram et al., 2015; Konopka et al., 2014; Peterson et al., 2008; Tarpey et al., 2017). However, studies on the relationship between a single bout of exercise and apoptosis are insufficient in comparison with those of chronic exercise on cardiac and skeletal muscle. In addition, it is pertinent to examine the effects of a single bout of exercise, since chronic exercise is a successive combination of responses to individual single bouts of exercise. Therefore, this study was performed to investigate the effects of a single bout of exercise on mitochondria-mediated apoptotic signaling in cardiac and skeletal muscle.
MATERIALS AND METHODS
Experimental design
Four-month-old Fischer 344 rats were randomly divided into 2 groups (n=10 per group): control group (CON) and a single bout of exercise group (EX). Cardiac (left ventricle) and skeletal muscles (e.g., soleus, type I fiber and white gastrocnemius, type IIb fiber) were collected from the respective groups and protein levels of mitochondria-mediated apoptotic signaling (e.g., Bax, Bcl-2, cytochrome c, and cleaved caspase-3) were measured via Western immunoblot analysis.
Animals and ethical approval
All procedures for animal experiments adhered to the stipulations of the National Institutes of Health and the guidelines of the Korean Academy of Medical Science. Rats were housed under controlled environmental conditions with constant illumination (12:12-hr light/dark) and room temperature (20°C±2°C). Additionally, food and water were made available to the rats ad libitum.
Exercise design
The treadmill exercise was performed following an adaptation period of about 10 min for a week. The single bout of EX received a single bout of treadmill exercise training at 20 m/min for 1 hr. The incline of this exercise was 0% and the intensity was approximately ~60% of the rats’ maximum oxygen uptake (VO2max), which corresponds with a moderate intensity (Schefer and Talan, 1996).
Western immunoblot analysis
The level of proteins involved in mitochondria-mediated apoptotic signaling was determined. Left ventricle, soleus, and white gastrocnemius tissues were collected and frozen immediately at −80°C. About 30 μg of tissues were homogenized with lysis buffer and centrifuged at 14,000 rpm for 20 min and 10 min, respectively. Protein levels were quantified by bicinchoninic acid assay method and concentration was confirmed by enzyme-linked immunosorbent assay Reader. Protein was denatured at 95°C for 5 min, separated on an sodium dodecyl sulfate-polyacrylamide gel with running buffer at 100 V for 2 hr, and then transferred onto a nitrocellulose membrane with transfer buffer at 170 mA for 1.5 hr. After Ponceau S staining, the membranes were blocked with 5% skimmed milk in Tris-buffered saline containing 0.1% Tween-20 at room temperature for 2 hr. The membranes were incubated overnight at 4°C with the following primary antibodies: GAPDH, Bax, Bcl-2, cytochrome c (Santa Cruz Biotechnology, Dallas, TX, USA) and cleaved caspase-3 (Cell Signaling Technology, Beverly, MA, USA). The membranes that reacted with the primary antibody were incubated at room temperature for 1 hr with horseradish peroxidase-conjugated anti-mouse (Vector Laboratories, Inc., Burlingame, CA, USA) for GAPDH, Bax, Bcl-2, and cytochrome c and anti-rabbit (Vector Laboratories, Inc.) for cleaved caspase-3. The bands were detected by using the enhanced chemiluminescence detection reagent kit (Thermo Fisher Scientific, Santa Clara, CA, USA) and expressed through Chemidoc (Bio-Rad, Hercules, CA, USA).
Statistical analysis
Data were presented as mean±standard error of the mean. An independent t-test was conducted to compare the effects of a single bout of exercise between groups. Significance level was set at 0.05. All graphs were produced using Prism 5 (GraphPad, La Jolla, CA, USA).
RESULTS
Effects of a single bout of exercise on Bax and Bcl-2 in cardiac and skeletal muscles
In cardiac and skeletal muscles the levels of Bax, a pro-apoptotic protein, were not significantly increased in EX compared with CON (Fig. 1). Also, the levels of Bcl-2, an antiapoptotic protein, were not significantly decreased in EX compared with CON in all tissues (Fig. 2), suggesting that the Bax/Bcl-2 ratio was not affected by a single bout of exercise (Fig. 3).
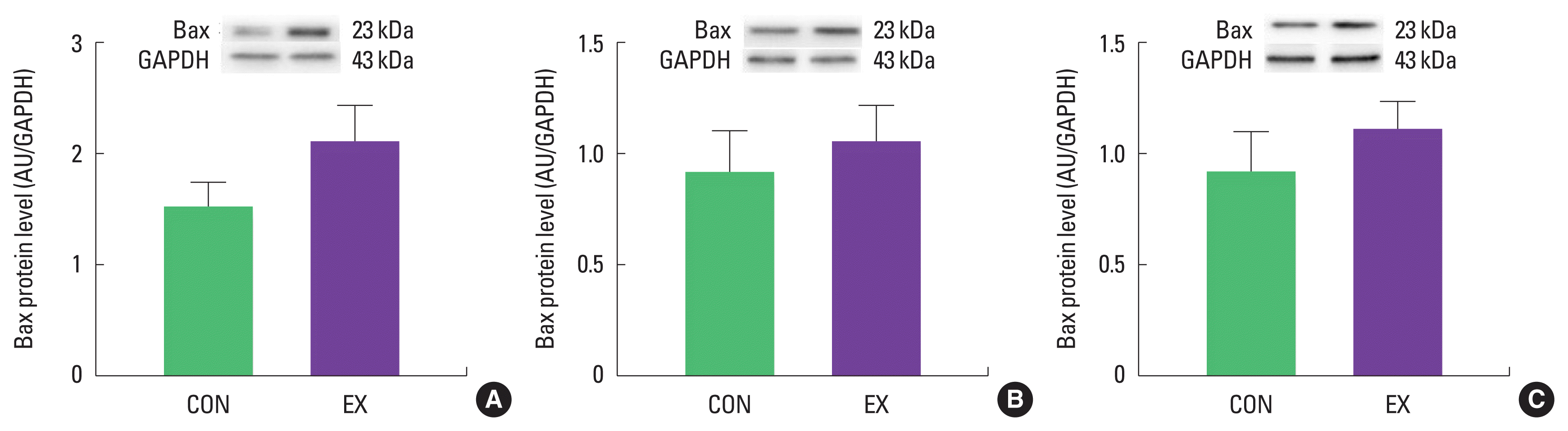
Effects of a single bout of exercise on Bax protein level in left ventricle (A), soleus (B), and white gastrocnemius (C). The data are shown as mean±standard error of the mean. CON, control group; EX, exercise group; AU, arbitrary unit; GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
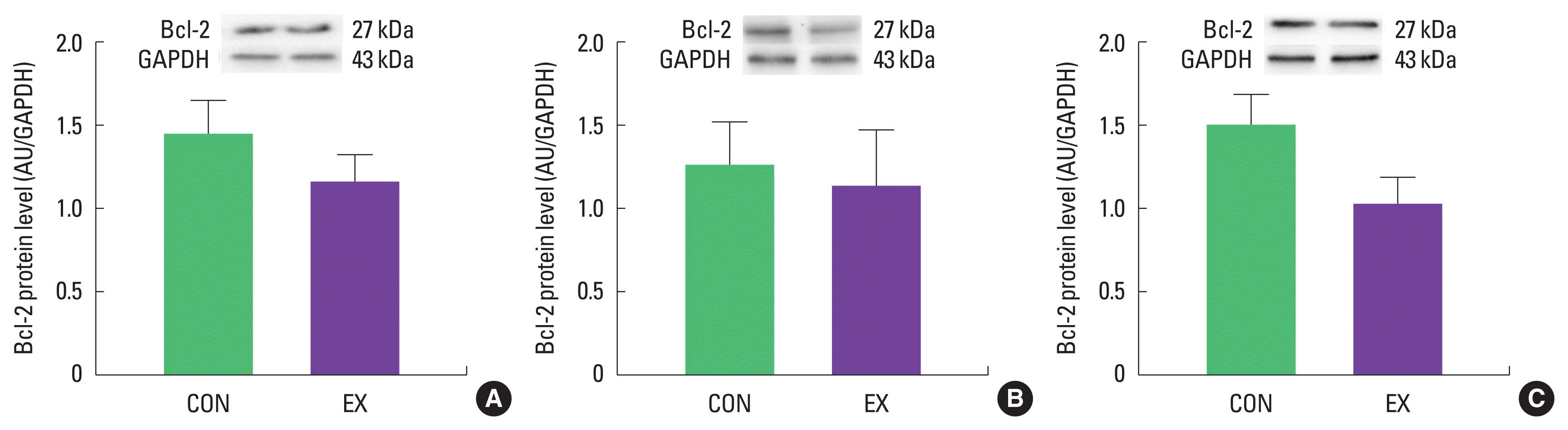
Effects of a single bout of exercise on Bcl-2 protein level in left ventricle (A), soleus (B), and white gastrocnemius (C). The data are shown as mean±standard error of the mean. CON, control group; EX, exercise group; AU, arbitrary unit; GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
Effects of a single bout exercise on mPTP, cytochrome c, and cleaved caspase-3 in cardiac and skeletal muscles
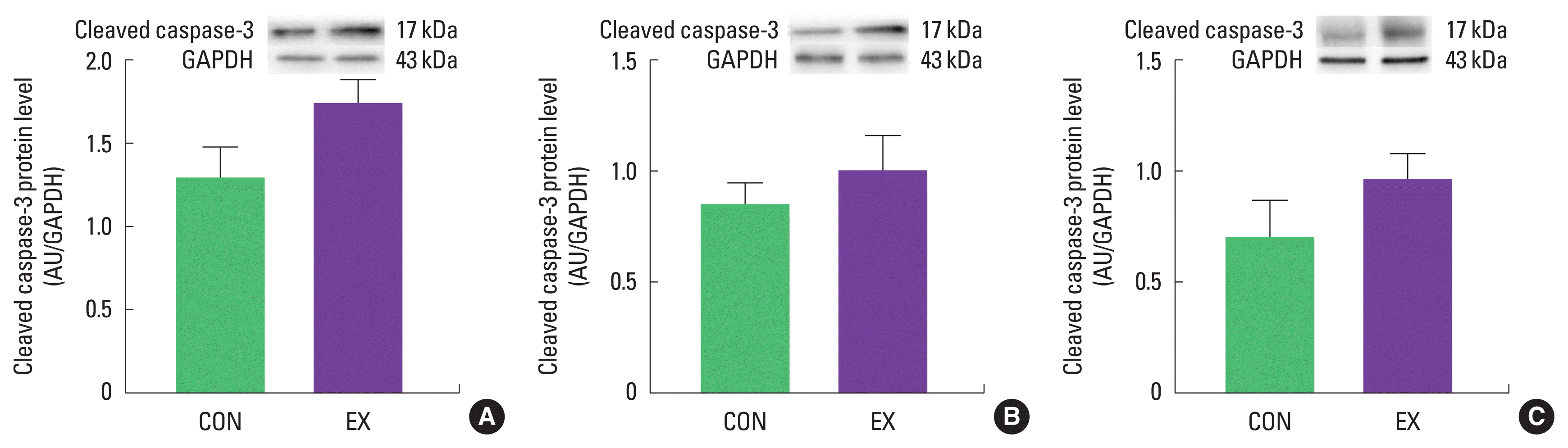
The mPTP opening sensitivity was significantly decreased in both cardiac and skeletal muscles by a single bout of exercise (P< 0.05) (Fig. 4). In contrast, protein levels of cytochrome c and cleaved caspase-3 were not different between CON and EX in cardiac and skeletal muscle (Figs. 5, 6).
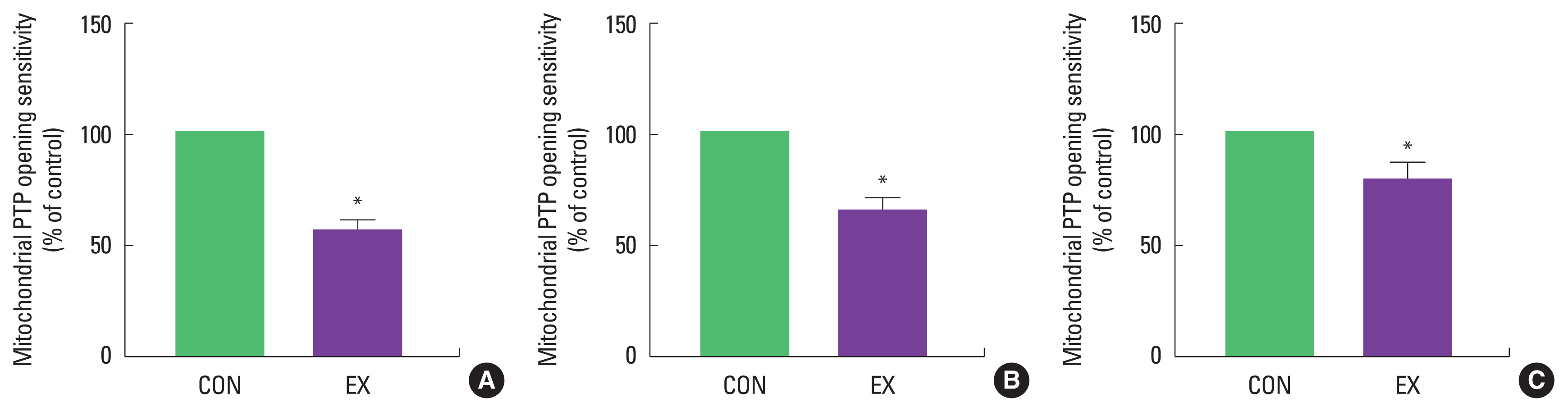
Effects of a single bout of exercise on mitochondrial permeability transition pore (PTP) opening sensitivity in left ventricle (A), soleus (B), and white gastrocnemius (C). The data are shown as mean±standard error of the mean. CON, control group; EX, exercise group; AU, arbitrary unit; GAPDH, glyceraldehyde-3-phosphate dehydrogenase. *P<0.05 compared to the control group.
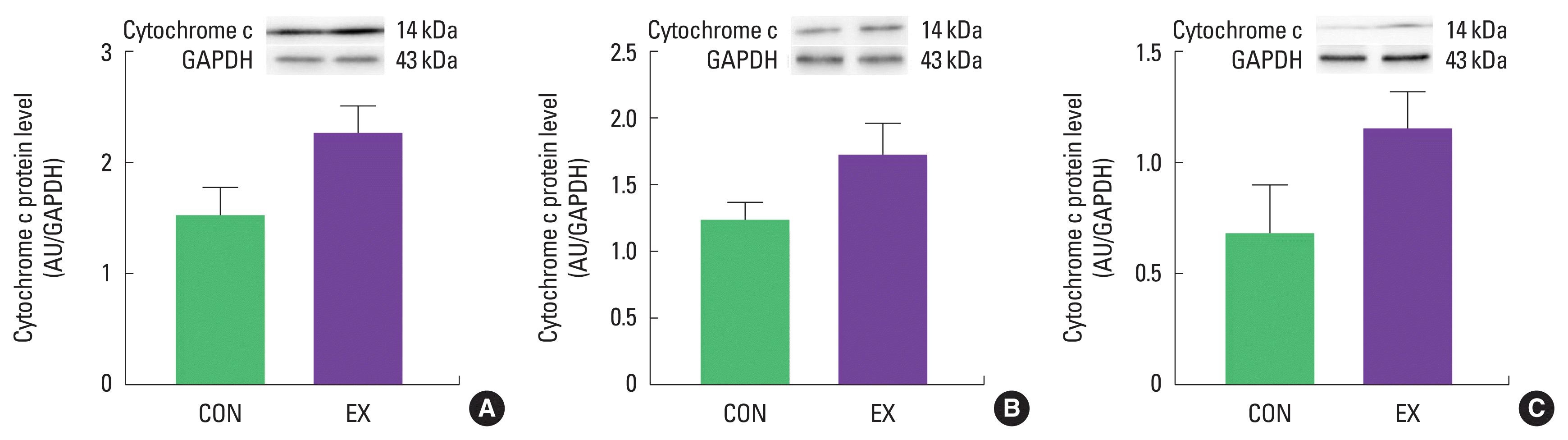
Effects of a single bout of exercise on cytochrome c protein level in left ventricle (A), soleus (B), and white gastrocnemius (C). The data are shown as mean± standard error of the mean. CON, control group; EX, exercise group; AU, arbitrary unit; GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
DISCUSSION
The purpose of this study was to investigate mitochondria-mediated apoptotic signaling responses to a single bout of exercise. We observed that a single bout of exercise significantly decreased mPTP opening sensitivity in all tissues (left ventricle, soleus, and white gastrocnemius). In contrast, a single bout of exercise did not show any statistical differences in Bax, Bcl-2, Bax/Bcl-2 ratio, cytochrome c, and cleaved caspase-3 between groups in both cardiac and skeletal muscles.
As mentioned before, mitochondria play a vital role in governing cellular events (survival and death) because they coordinate a process known as the mitochondria-mediated apoptotic pathway (Quadrilatero et al., 2011). Considering that apoptosis is mainly induced by mtROS and mPTP opening (Redza-Dutordoir and Averill-Bates, 2016; Xiong et al., 2014), our findings lead us to believe that a single bout of exercise may be related to mitochondria-mediated apoptotic signaling, because we observed that mitochondrial H2O2 emission in the white gastrocnemius (Yoo et al., 2019) was increased and the mPTP opening of all tissues was decreased by a single bout of exercise. mtROS can lead to the release of pro-apoptotic proteins such as Bax into the cytoplasm, consequently inducing increased mPTP opening, caspase-9, cleaved caspase-3, and DNA fragmentation (Heo et al., 2017; Xiong et al., 2014). However, in the current study, a single bout of exercise showed a tendency (a) to increase the protein levels of Bax, cytochrome c, and cleaved caspase-3; (b) to decrease Bcl-2 protein levels; (c) to increase the Bax/Bcl-2 ratio in all tissues, albeit without statistical significance. The observed data are not in agreement with previous studies. Contrary to our findings, a few studies have reported significant apoptotic markers in relation to a single bout of exercise (Boroujerdi and Rahimi, 2011; Lagranha et al., 2004; Park et al., 2011; Quadrilatero and Hoffman-Goetz, 2005; Rahimi et al., 2018; Sharafi and Rahimi, 2012). Indeed, intense acute resistance exercise resulted in an increase in pro-apoptotic protein, Bax and Bax/Bcl-2 ratio in humans (Rahimi et al., 2018).
Moreover, it was demonstrated that pro-apoptotic proteins increased following strenuous acute exercise in human (Boroujerdi and Rahimi, 2011; Park et al., 2011; Sharafi and Rahimi, 2012) and animal (Lagranha et al., 2004; Quadrilatero and Hoffman-Goetz, 2005) studies. We conjecture that these results may be due to the intensity of exercise. The exercise intensity we set in this study is moderate; ~60% of VO2max. However, the intensity of exercise utilized in previous studies is high, ranging from ~70% (Park et al., 2011) to ~85% (Lagranha et al., 2004) of VO2max. Also, according to Syu et al. (2011), acute severe exercise led to increased oxidative stress and apoptosis but chronic moderate exercise delayed apoptosis. Thus, we believe that a single bout of moderate exercise is insufficient to make a significant difference, but further study is needed to provide a more mechanistic understanding of the apoptosis pathway. One study reported that low intensity but a long duration of acute exhaustive swimming exercise significantly induced an increase in mRNA expression of Bcl-2 and a decrease in activity of caspase-3 (Sun et al., 2016). Moreover, studies of apoptosis in specific tissues are also needed because, while numerous studies have examined blood serum levels, few have also sought to identify apoptosis factors in specific tissues.
In conclusion, a single bout of exercise did not result in observable changes in Bax, Bcl-2, Bax/Bcl-2 ratio, cytochrome c, and cleaved caspase-3 in cardiac and skeletal muscle. However, a significant decrease in mPTP opening sensitivity in both cardiac and skeletal muscle was observed. Further research is necessary to provide a more mechanistic understanding of mitochondria-mediated apoptosis in the future.
ACKNOWLEDGMENTS
This work was supported by the Inha University Research Grant (2018).
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.