Influence to high-intensity intermittent and moderate-intensity continuous exercise on indices of cardio-inflammatory health in men
Article information
Abstract
The aim of this study was to evaluate the influence to acute exercises performed in different intensities with volume equalized (5 km) on indices of cardio-inflammatory health. Twelve physically active male subjects (age, 23.22±5.47 years; height, 174.75±5.80 m; weight, 75.13±6.61 kg; maximal oxygen uptake, 52.92 mL/kg/min), after determination of peak oxygen uptake (VO2Peak) and the speed associated with VO2Peak (sVO2Peak), completed two randomly experimental trials: high-intensity intermittent exercise (HIIE: 1:1 at 100% sVO2Peak) and moderate-intensity continuous exercise (MICE: 70% sVO2Peak). Brain-derived neurotrophic factor (BDNF), adiponectin and plasminogen inhibitor-1 (PAI-1) data were analyzed pre, immediately, and 60 min after the exercise session. Statistical analysis comparisons between moments and between HIIE and MICE were performed using a mixed model and statistical and significance was set at <5%. PAI-1 presented an effect for time from pre to immediately after exercise moment (P<0.018) and from immediately to 60 min after exercise moment (P<0.001) only in MICE. BDNF presented an effect for time from pre to immediately after exercise to HIIE (P<0.022) and from immediately to 60 min after exercise to MICE (P<0.034). HIIE promotes BDNF increase and that there is negative correlation between PAI-1 concentrations and BDNF in both protocols in healthy sportsmen, favoring an anti-atherogenic profile.
INTRODUCTION
Cardiovascular diseases, such as atherosclerosis and coronary artery disease, are the main cause of death worldwide (McPherson et al., 2015; Pedersen, 2009; Rosamond et al., 2008), and are closely related to alterations on lipids metabolism, endothelial function, and inflammatory mediators (Hotamisligil et al., 1996; Lira et al., 2012). These diseases influence signaling proteins levels in circulatory blood, such as, adiponectin reduction and increased production and release of plasminogen inhibitor-1 (PAI-1) (Jovin et al., 2003; Kohler and Grant, 2000; Punyadeera et al., 2005).
Regarding the last outcome, PAI-1 has been receiving attention due its relationship to the atherosclerosis process and coronary artery disease, which can be mediated by increased low-density lipoprotein levels (Kantor et al., 1987; Lira et al., 2009). On the other hand, adiponectin has been recognized as anti-inflammatory and antiatherogenic cytokine, and is related to fat oxidation (Kraemer et al., 2003; Lira et al., 2012).
Recently, brain-derived neurotrophic factor (BDNF), that is a member of neurotrophin family involved in neuroprotection and neurogenesis with direct consequences on the cognition and mood parameters, has been linked with metabolism challenge and cardiovascular health (Kaess et al., 2015; McPherson et al., 2015). Kaess et al. (2015) showed that higher BDNF levels are associated with decreased risk of cardiovascular disease, and concluded in their studies that BDNF levels can be considered as heart failure biomarker. Furthermore, Prigent-Tessier et al. (2013) in their study with rats, showed the negative and positive impacts of high blood pressure and physical training, respectively, on the relationship of endothelial function with BDNF.
Classical moderate-intensity continuous exercise (MICE) is accepted as antiatherogenic and anti-inflammatory, since it increases reverse cholesterol transport toward high-density lipoprotein cholesterol particles, and promote clearance of cholesterol ester from tissues and lipoproteins (Gupta et al., 1993; Vaisberg et al., 2012). Moreover, MICE improve blood circulation in central nervous system and stimulate neurotrophin production. Recently, high-intensity intermittent exercise (HIIE) has been shown to induce similar or higher improvement than MICE exercise (Gibala et al., 2012). Williams et al. (2013) verified the similarity between MICE and HIIE in increasing factors linked to anti-inflammatory status. However, exercise volume was significantly different between sessions, which likely influenced their response.
Few studies investigated the effects of HIIE on indices cardio-inflammatory, when the session exercise volume is equalized. Therefore, given that it is unknown whether HIIE and MICE present differences in inflammatory and atherosclerotic biomarkers, the present study aimed to compare serum BDNF, PAI-1, and adiponectin concentrations after HIIE and MICE acute exercise with an equal volume of work (5 km).
The present study would like to compare atherosclerotic markers responses after acute exercises performed in different intensities with volume equalized (5 km), and thus understand the importance of acutely measuring parameters such as BDNF, PAI-1 and adiponectin to understand the long-term impact.
MATERIALS AND METHODS
Subjects
Twelve male sportsmen volunteered for the present study and all subjects presented conditions to be submitted to the exercises, and the procedures were in accordance to ethical standards of the Research Ethics Committee for studies involving human participants of the University of the State of São Paulo (UNESP) – Campus Presidente Prudente/Brazil and the ethical standards of World Medical Association Declaration of Helsinki. The subjects were informed about the risk and benefits to participate to the present study, and assigned the informed consent.
Procedures
After an incremental test to determine peak oxygen consumption (VO2peak) and the speed associated with it (sVO2peak). Before exercise, subjects warmed-up consisting of a running for 5 min at 50% of sVO2peak at 1% inclination and performed randomically two exercise sessions. In the HIIE, subjects performed 1-min at sVO2peak with 1-min of passive rest while the MICE was performed continuously at 70% of VO2peak, both with 5-km volume. The exercise sessions were separately by at least 72 hr, were performed in the same time of day and a temperature between 22°C±2°C. The subjects were instructed to not execute strenuous exercise for at least 24 hr prior each session or to ingest stimulants (tea, coffee, chocolate).
Maximal endurance running test
The incremental test was conducted on a treadmill (Inbramed MASTER CI, Inbrasport, Porto Alegre, Brazil) with initial speed of 8 km/hr. The speed increase 1 km/hr each 2 min until exhaustion. The subjects were verbal encouraged during the test. The oxygen uptake was analyzed by Quark PFT (Cosmed, Rome, Italy). The highest VO2 observed during test was assumed as VO2peak. The sVO2peak was considered as the last speed of incremental test. If subject did not complete the stage, the speed was adjusted as proposed by Kuipers et al. (1985): sVO2peak=speed of final complete stage+[(time, in seconds, remaining at the final incomplete stage/120 sec) ×1 km/hr].
To control whether subjects gave they maximal effort, they had to attain three or more of the following indexes: 1, VO2 plateau (≤150 mL/min); 2, the rating of perceived exertion≥18; 3, attainment of the percentage of the age-predicted maximal heart rate within ±5 beats/min; 4, respiratory exchange ratio ≥1.10 (Howley et al., 1995).
HIIE and MICE sessions
For both exercise trials, the subjects performed a warm-up consisting of a running at 50% of sVO2peak for five minutes at 1% inclination. The HIIE was performed intermittently with subjects running on a treadmill for one minute at 100% of sVO2Peak, interspersed by one minute of passive recovery (without exercise) until they had complete 5 km. The MICE consisted of a continuous 5-km run on the treadmill at 70% of sVO2Peak.
Blood sampling and analyses
The blood samples were collected at rest, and immediately, and 60 min after exercise sessions for both HIIE and MICE. The blood samples (15 mL) were immediately allocated into vacutainer tubes (Becton Dickinson, BD, Juiz de Fora, Brazil) containing ethylenediaminetetraacetic acid for plasma separation and into dry vacutainer tubes for serum separation. The tubes were centrifuged at 3.500 g for 15 min at 4°C. Plasma and serum samples were stored at −20°C until analysis. PAI-1, BDNF, and adiponectin levels were assessed using enzyme-linked immunosorbent assay commercial kits (R&D Systems, Minneapolis, MN, USA).
Statistical analysis
The data normality was verified using the Shapiro-Wilk test and for each variable mean and standard deviations were calculated. Mixed models for repeated measures (one factor) were used to examine differences in blood variables for each condition with regard to response time. The Tukey test was used post hoc when differences were found. The significance level was set at 5%.
Pearson correlation was used to analyze the changes in the concentrations in adiponectin, BDNF and PAI-1 (Δ=[60-min postexercise]–[rest]). The data was analyzed using the SAS 9.3 (SAS Institute Inc., Cary, NC, USA).
RESULTS
Table 1 presents the mean values of both protocols of this study.
For the MICE exercise there was a time effect PAI (F2, 15.8=12.0, P<0.001) with higher values immediately post-exercise than at rest (P=0.018) and 60-min postexercise (Fig. 1A) (P<0.001).
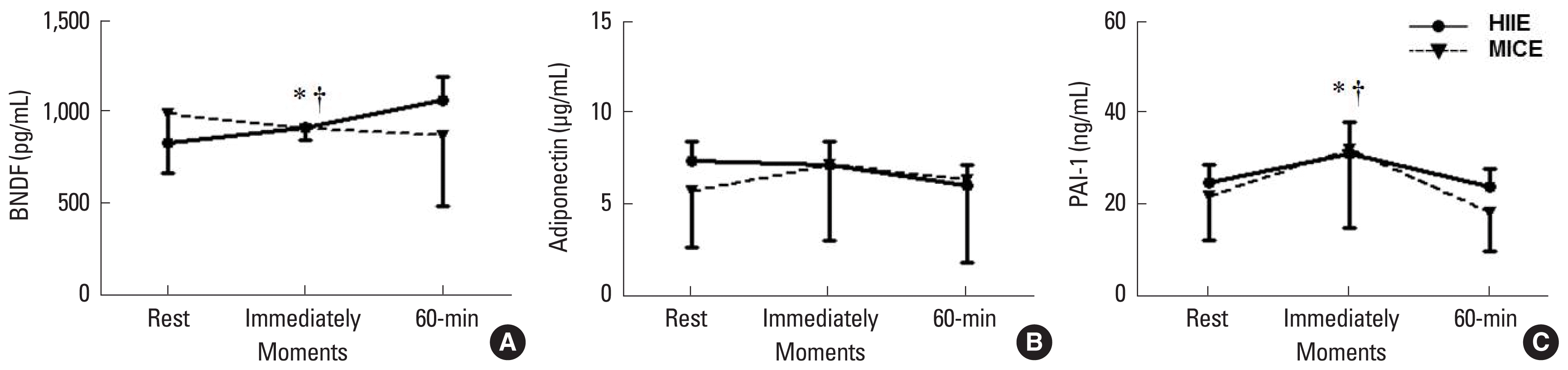
Effects of MICE and HIIE upon BDNF (A), adiponectin (B), and PAI-1 (C). Data are mean±standard deviation. MICE, moderate-intensity continuous exercise; HIIE, high-intensity intermittent exercise; BDNF, brain-derived neurotrophic factor; PAI-1, plasminogen activator inhibitor-1. *P<0.05 in relation rest moment. †P<0.05 in relation 60-min moment.
For HIIE there was a time effect for BDNF (F2, 6.69=6.27; P= 0.036) with higher values immediately and 60-min postexercise than at rest (P=0.022) (Fig. 1B).
There was a not interaction effect to adiponectin (Fig. 1C).
Aiming to better understanding of relationship between atherogenic and antiatherogenic markers we sought to use Pearson correlation to verify a possible association among these indicators after exercise protocols. Thus, it was revealed that there was a tendency to positive relationship between ΔAdiponectin and ΔBDNF in MICE (rho=0.472, P=0.121), but not in HIIE (rho= −0.009, P=0.978). Interestingly, there was a tendency to negative relationship between ΔPAI-1 and ΔBDNF in the HIIE (rho= −0.417, P=0.177) as well MICE (rho=−0.516, P=0.086), indicating that the increase in BDNF levels seen in this condition was associated with lower PAI-1 concentrations (Table 2).
DISCUSSION
The novelty of this study was the investigation of the effects of exercise with an equal volume of work in two different intensities on atherogenic (PAI-1) and antiatherogenic response (adiponectin and BDNF), which are cardiovascular health biomarkers. The main finding of this study was there is a divergent response of BDNF levels following MICE and HIIE. In addition, we demonstrated that PAI-1 enhanced immediately after exercise regarding rest in both conditions.
PAI-1 is important in balancing the fibrinolytic processes by inhibiting the tissue plasminogen activator action (Cesari et al., 2010). However, when PAI-1 circulates at high levels, it favors the increase of thrombosis and, thereby, cardiovascular risk (Cesari et al., 2010). As seen in Fig. 1A, in MICE, PAI-1 concentrations increased from rest to immediately after exercise (P<0.018) and decreased throughout 60 min (P<0.001). These responses are in agreement with previous studies (El-Sayed, 1996). Kahraman et al. (2011) verified changes in the fibrinolytic system in young subjects submitted to MICE (50%–65% VO2Peak) for 60 min and PAI-1 responses were found to be similar to the present study. Szymanski and Pate (1994) compare the relationship between acute submaximal exercises (80% VO2max) on fibrinolytic responses, and found that PAI-1 activity was decreased postexercise when compared to the basal value.
On the other hand, the HIIE group did not present alterations in PAI-1 concentrations. To our knowledge, this is the first study that tested the acute effect of HIIE on PAI-1 and the understanding of the possible mechanisms involved in this type of exercise are limited. However, Kahraman et al. (2011) reported that in heavy exercise fibrinolysis and coagulation are activated, probably by a neutralization response, so the fibrin could have inhibited production by fibrinolytic mechanisms (Weiss et al., 1998). Indeed, more studies are needed to clarify the dynamic of PAI-1 concentrations in HIIE.
In the present study, no alterations were found regarding adiponectin levels in both protocol exercises. Our results are compatible with previous reports (Magkos, 2009; Williams et al., 2013). Magkos et al. (2006) suggest that enhance of adiponectin levels occurs in long-term, and these findings are reinforced by literature (Moghadasi et al., 2012; Simpson and Singh, 2008), but not as an acute response. However, there are conflict results (Kraemer et al., 2003). An increase in adiponectin concentrations was found after 30 min of recovery when compared with rest moment in high-intensity rowing exercise (Jürimäe et al., 2005), indicates that HIIE can modulate adiponectin secretion. However, these studies failed to analyze the time-course of adiponectin after an exercise session.
The main finding of this study was about BDNF, which behaved differently between the two protocols. In HIIE, the BDNF level increased immediately after exercise, while in MICE, it reduced at 60-min recovery period. This result is relevant, since HIIE seems to be more favourable to antiatherogenic acute response than MICE in protocols with equal volume of exercise. BDNF is related with neuroprotection and neurogenesis, cognition function, mood, metabolism challenge and cardiovascular health (Kaess et al., 2015). Thus, it is desirable to find better methods to stimulate its production.
Our results are not entirely similar to those found in the literature. Regarding HIIE, it was found only the study of Saucedo Marquez et al. (2015) that verified BDNF behaviour in humans submitted to HIIE. Saucedo Marquez et al. (2015) showed that BDNF concentrations did not reduce during recovery period. Most studies in the literature involving BDNF and exercise refer to moderate intensity aerobic exercise. The authors attributed this response to lifestyle and/or BDNF polymorphism that affect 50% of Japanese population. However, in the present study, BDNF levels decreased after 60 min after MICE. A justification for this difference in relation to others studies may be the session duration, since the studies that showed an increase of BDNF immediately after exercise there are low (20–30 min) while in the present study the average time of MICE in our study was beyond 50 min.
Furthermore, the nonincrease in MICE as well as the increase in HIIE in BDNF levels showed a tendency to negative correlation with PAI-1. However, these are only speculations that need to be tested.
The limitation of this study is related to the lack of subjects suffering from chronic diseases in which the changes in variables could be assessed and, thus, would be more effective. In summary, we demonstrated that HIIE seems better to improve BDNF levels, and that there is negative correlation between PAI-1 concentrations and BDNF in both protocols in healthy sportsmen.
ACKNOWLEDGMENTS
This work was supported by the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, Brazil) under Grant No. 2013/25310-2.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.

