AbstractThe main purpose of this study was to suggest a solution for inflammation caused by urban daily life based on supercritical extraction of Chamaecyparis obtusa Endl. (C. obtusa). The leaves, cone, wood, and bark of C. obtusa were separately extracted with supercritical extraction. Alpha-pinene, anti-inflammatory active compound of C. obtusa, was analyzed by electrospray ionization-mass spectrometry, gas chromatography, and gas chromatography mass spectrometry from each part of extracts. As a result of the reduction rate of nitric oxide in RAW 264.7 cells, the wood extract exhibited the highest reduction rate was about 45% among extracts from four part of C. obtusa. The wood extract from C. obtusa can be developed as a good anti-inflammatory natural raw material for natural product industry.
INTRODUCTIONModern people increased interesting in natural materials with various functions as the interest of the environment and health. Among the various natural products, Chamaecyparis obtusa Endl. (C. obtusa) has a positive image for Korean people and it is easy to find and without any discomfort when using. Already research studies revealed that C. obtusa has anti-inflammatory effect (Kim et al., 2015; Rufino et al., 2014). Inflammation is on action for a variety of reason (Lee et al., 2017; Park et al., 2018; Song et al., 2017). If inflammation action in the skin, it can be various reactions such as increase of blood flow blood flow increase, cell metabolism enhancement, vasodilation to defend and recover (Ferrero-Miliani et al., 2007). The proper production of mediators such as interleukin-4, interleukin-6, and nitric oxide (NO) produced by inflammation is beneficial to the body, but overproduction is involved in rheumatoid arthritis and bacterial sepsis (Dinarello, 1997; Palladino et al., 2003). Therefore, in this study, we tried to examine the components of each extracts and anti-inflammatory effect of extract from each part of C. obtusa with component. In addition, it was the supercritical extraction, which is a natural extraction method. It was analyzed whether extracts from C. obtusa using the natural extraction method have inflammation related components and investigated effect of anti-inflammatory on RAW264.7 cells.
MATERIALS AND METHODSSupercritical fluid extractionThe required pretreated C. obtusa was charged into the extractor (① of Fig. 1). The supercritical carbon dioxide was supplied into the extractor via the CO2 circulation pump (⑤ of Fig. 1). The supercritical condition was maintained by controlling both of pressure and temperature through preheater (⑧ of Fig. 1). After setting up of the required conditions extraction can commence. After the supercritical CO2 extraction, supercritical CO2 was separated in the separator (②, ③ of Fig. 1), where the gasified CO2 was separated from the product. The gaseous CO2 was transferred to cooling unit to be liquified and collected to the working tank (④ of Fig. 1). The extract from C. obtusa was withdrawn by means of pressure reduction and vaporization in the separator. The unloaded gaseous CO2 was condensed afterwards and fed back to the working tank. The CO2 circulation continues. The reuse of recycle CO2 made this process as the continuous operation until the extraction was over (Fig. 1).
Fatty acid analysisThe fatty acids of extract from C. obtusa were analyzed by gas chromatography. Using HP 5890 system (HP 19251A, Hewlett-Packard, Palo Alto, CA, USA) was used and flame ionization detector was used. The BF3 method (Dawidowicz and Thompson, 1971) was modified and the fatty acid standard (Sigma, St. Louis, MO, USA), used in the methylation, was C14, C16, C18, C18:1, C18:2. The extracts used 2N-KOH/MeOH for methylation. The column used Supelco SP-2340 Fused Silica Capillary Column (30 m, 0.25 mm [in side diameter], 0.20 μm [Film]) and the oven condition was 170°C→2°C/min→220°C 2 min hold, the detector and the injector temperature set at 230°C.
Electrospray ionization-mass spectrometryMass spectrometry analysis was occurred using electrospray ionization-mass spectrometry (ESI-MS) by Ruth’s method for the analysis of the amount of index substance extracted with supercritical extraction time. The single quadrupole system (API-150EX, Applied Biosystem, Foster, CA, USA) was used for the analysis and the ion source was used in the ESI mode. The extracts were anaylzed at a concentration of 100 mg/L in chloroform: methanol:2-propanol (1:2:4) with 7.5 mM ammonium acetate and filtered using a 0.25-μm syringe filter for injection. The infusion solvent was 50% methanol and the flow rate was 10 μL/min.
Solid phase microextraction methodThe solid phase microextraction method (SPME) is a method of separating and concentrating a small amount of analytes of a sample using an adsorbent. It was the process by which the fiber and analytes of sample were adsorbed and then analyzed in a separator of the analyzer. The SPME fiber used was SUPELCO 65 μm PDMS/DVB (57310-U, Supelco, Bellefonte, PA, USA) and temperature was room temperature, adsorption time was 40 min, and desorption time was 5 min. The column used DB-FFAP 60 m× 0.25 mm (ID)×0.5 μm (Film) (Agilent, Santa Clara, CA, USA) and the oven condition was 60°C (5 min hold)→2°C/min→ 220°C (60 min hold), The detector and the injector temperature set at 240°C.
Nitric oxide assayCulture 5×104 cells/well RAW264.7 cells from Korean Cell Line Bank in the 96 well plate. After 24 hr, cells treated with 1 ppm lipopolysaccharides and different concentration of emulsion which made with Dulbecco’s modified eagle medium in the media. (0, 1, 5, 10, 50, 100, 200 ppm) After 24 hr, sampling 100-μL supernatant media, adding 100-μL Griess reagent (50-μL sulfanilamide+ 50-μL N-1-napthylenediamide dihydrochloride solution). Incubate for 10 min at room temperature. Determine the absorbance of each well at the 490 nm.
RESULTSSupercritical fluid extractionAs the temperature increased, the extraction efficiency was also increased. The highest efficiency was obtained at 60° (Fig. 2). The extraction time was determined to be 60 min, based on the 90% intensity of α-pinene of marker component on ESI-MS (Fig. 3). In case of bark was too viscous to be extracted with time, so it was extracted at the same conditions as another part at 350 bar and 60°C for 60 min.
Fatty acid componentFatty acid (myristic acid [C14], palmitic acid [C16], stearic acid [C18], oleic acid [C18:1], linoleic acid [C18:2], linolenic acid [C18:3]) in supercritical extracts from C. obtusa were analyzed by GC. As in Fig. 4, the fatty acid composition has a big difference in each part extract except myristic acid. The most difference was that palmitic acid was not analyzed in wood extracts, while leaf extracts showed more than half the composition (55.4%). On the contrary, oleic acid showed the highest composition of 37.5% in wood extract but 5.1% in leaf extract. Except for leaf extract, oleic acid content was high in the all extracts. Especially, the content in bark was about half. Materials for extraction were collected from the same tree, but fatty acid compositions were not same (Table 1).
GC/MS analysisAs a result of GC/MS analysis for each component of four parts, the kind of similar volatile components and proportion of components in leaves and cone was analyzed (Table 2). The most number of volatile component was analyzed extract from bark of C. obtusa. Alpha-pinene taken as the indicator material was analyzed at the highest rate in extract from wood. In addition, the ratio of α-pinene analyzed by GC/MS was analyzed at the same ratio as that of intensity on ESI-MS for optimization of extraction time in supercritical extraction.
DL-limonene, which was commonly analyzed at three parts (leaves, cone, wood) has been reported to physiologically active substance effective against antifungal and aflatoxin and myrcene has been reported to effective substance of anti-inflammatory (Rufino et al., 2014; Singh et al., 2010; Tavares et al., 2010).
Effects of extracts from C. obtusa and α-pinene on NOAs shown in Fig. 5, the NO depression results of the extracts from four parts of C. obtusa showed that NO production was reduced in all extracts from four parts. NO inhibition was highest in wood extracts and lowest in extract from leaves. It was shown that NO production was reduced as the ratio of α-pinene analyzed by GC/MS was high. Among extract from four parts, highest difference was observed at 200 ppm which has not occurred cytotoxicity And NO production inhibition of the α-pinene standard was about 51% at 200 ppm (Fig. 6). NO inhibition rate of α-pinene was more than 6% wood extract, which had the highest inhibition rate of NO production among four parts of extract. The result of analysis and the NO inhibition rate was shown that the NO inhibition rate was proportional to the concentration of α-pinene which was marker a component.
DISCUSSIONConponent of essential oil from C. obtusa was almost terpenoid. The main component of C. obtusa in Korea was known as monoterpenes and sesquiterpenes. It is a volatile oil extracted from the leaves, roots, stems and flowers that has antibacterial effects of sabinene, limonene, bornly, acetate, borneol, α-terpineol, and elemol have been known. The hinokitio and thujaplicin compounds in essential oils obtained from wood have physiological activities such as antibacterial, antioxidant, germination inhibition of plants, and freshness maintenance of plants. These substances are effective against inflammation and stimulation of humans. For example, in the cosmetics industry, a lot of chemicals were contained in the manufacture of cosmetics such as surfactants, preservatives, synthetic fragrances, etc. which can cause irritation when these contact with the skin for a long time. Many anti-irritants inhibit the stimulation from these ingredients, but it was necessary to develop safe and effective from natural materials that could protect the skin from various factors and relieve the irritation (Held et al., 1999). Among the various natural products, C. obtusa has a positive image to Korean people and it is easy to find and without any discomfort when using. on this study extracts from C. obtusa by supercritical extraction could be used as an anti-inflammatory substance by various inflammatory reactions in the natural product industry.
REFERENCESDawidowicz EA, Thompson TE. Artifacts produced by boron trifluoride methanolysis of a synthetic lecithin containing cyclopropane fatty acids (1–2-dihydrosterculoyl-3-sn-phosphatidylcholine). J Lipid Res. 1971;12:636–637.
Dinarello CA. Proinflammatory and anti-inflammatory cytokines as mediators in the pathogenesis of septic shock. Chest. 1997;112:6 Suppl. 321S–329S.
Ferrero-Miliani L, Nielsen OH, Andersen PS, Girardin SE. Chronic inflammation: importance of NOD2 and NALP3 in interleukin-1beta generation. Clin Exp Immunol. 2007;147:227–235.
Held E, Sveinsdóttir S, Agner T. Effect of long-term use of moisturizer on skin hydration, barrier function and susceptibility to irritants. Acta Derm Venereol. 1999;79:49–51.
Kim DS, Lee HJ, Jeon YD, Han YH, Kee JY, Kim HJ, Shin HJ, Kang J, Lee BS, Kim SH, Kim SJ, Park SH, Choi BM, Park SJ, Um JY, Hong SH. Alpha-pinene exhibits anti-inflammatory activity through the suppression of MAPKs and the NF-κB pathway in mouse peritoneal macrophages. Am J Chin Med. 2015;43:731–742.
Lee T, Lim US, Kang DH, Jung HD, Kim H, Choi BH, Kang JH, Yoon SM, Park CS. Near-normalized gene expression profiles in bladder with detrusor overactivity in rats with bladder outlet obstruction after deobstruction. Int Neurourol J. 2017;21:247–258.
Palladino MA, Bahjat FR, Theodorakis EA, Moldawer LL. Anti-TNF-alpha therapies: the next generation. Nat Rev Drug Discov. 2003;2:736–746.
Park JS, Jin MH, Hong CH. Neurologic mechanisms underlying voiding dysfunction due to prostatitis in a rat model of nonbacterial prostatic inflammation. Int Neurourol J. 2018;22:90–98.
Rufino AT, Ribeiro M, Judas F, Salgueiro L, Lopes MC, Cavaleiro C, Mendes AF. Anti-inflammatory and chondroprotective activity of (+)-α-pinene: structural and enantiomeric selectivity. J Nat Prod. 2014;77:264–269.
Fig. 1The process flow diagram for the supercritical extraction from Chamaecyparis obtusa. The conditions used for the extraction from C. obtusa were pressure of 350 bar, temperatures of 40°C, 50°C, 60°C, flow of 145 mL/min, particle size of 800–1,000 μm. 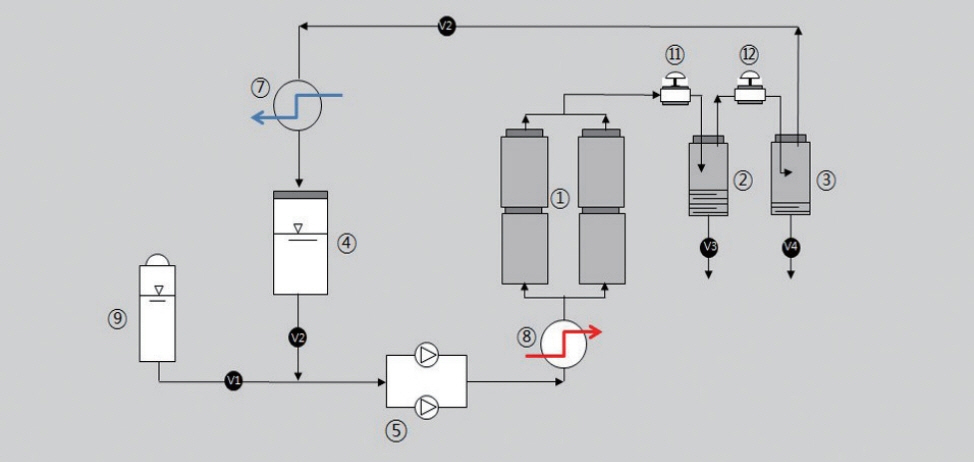
Fig. 2The effect of temperature on the extraction rate from Chamaecyparis obtusa. Supercritical CO2 flow was 145 mL/min, extract time was 120 min, extractions were treated at 40°C, 50°C, 60°C. Leaves (A), woods (B), cones (C). 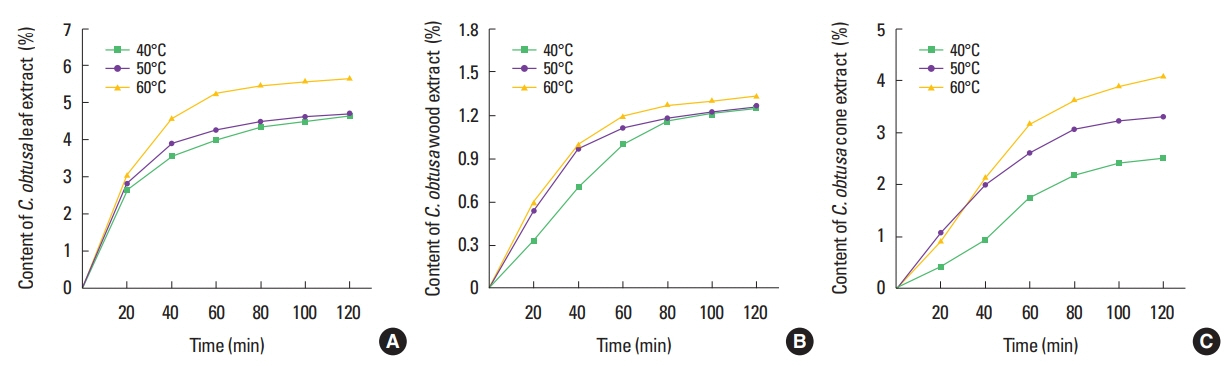
Fig. 3Intensity comparison of α-pinene from Chamaecyparis obtusa extracts. Analysis by electro spray ionization mode of mass spectrometry. (A) Peak in the box was α-pinene. (B) Alpha-pinene peak intensity was highest on wood extracts. 
Fig. 4Chromatogram of extracts from Chamaecyparis obtusa. Chromatogram of extracts from bark. Gas chromatography conditions: 170°C to 220°C at 2°C/min; flame ionization detector, 230°C; split ratio, 20:1. 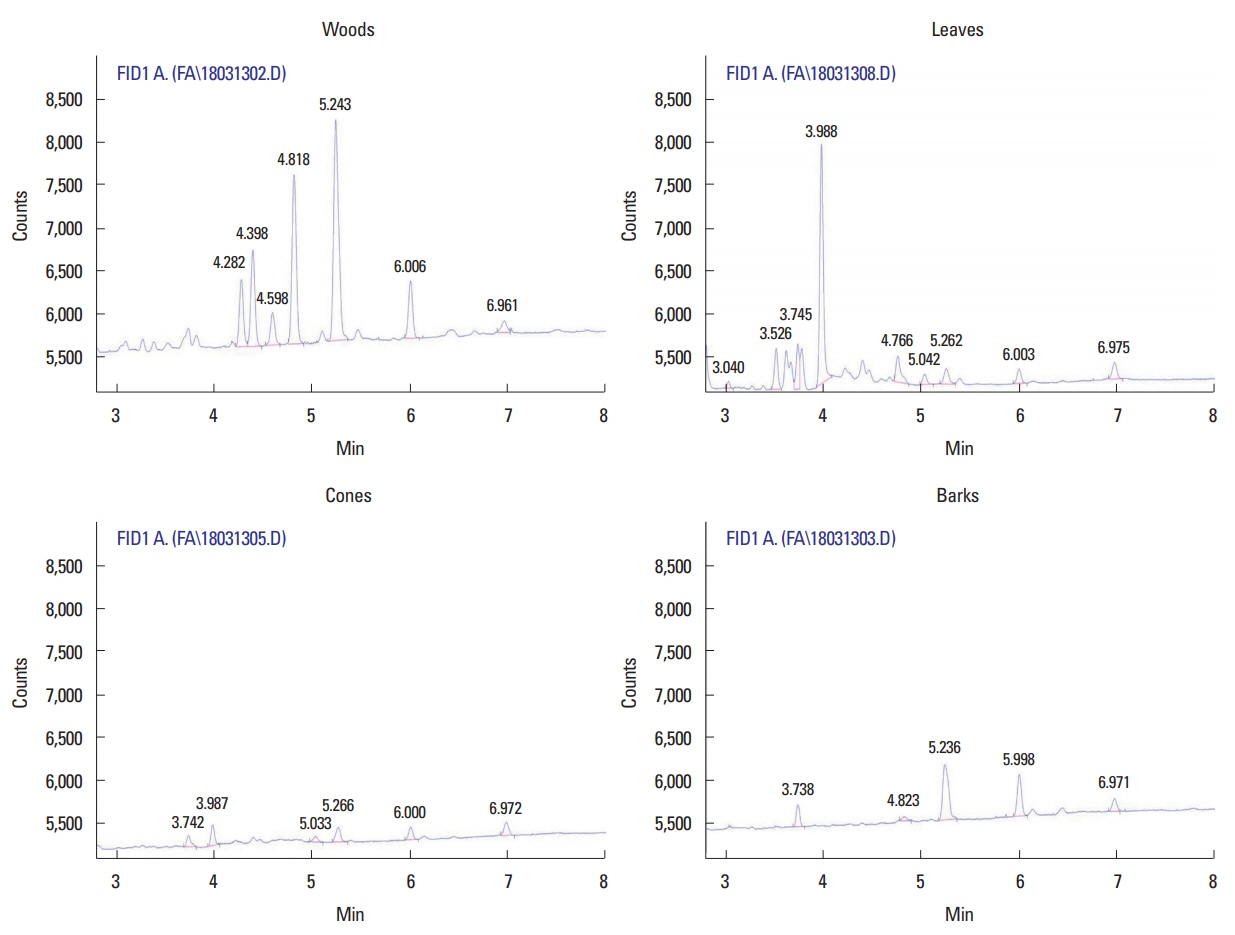
Fig. 5Inhibition of nitric oxide production on RAW 264.7 cells by extracts from Chamaecyparis obtusa. Nitric oxide (NO) production was activated by lipopolysaccharide in RAW 264.7 cells. *P<0.05, **P<0.01, ***P<0.001 vs. control. The data were obtained from 3 independent experiments. 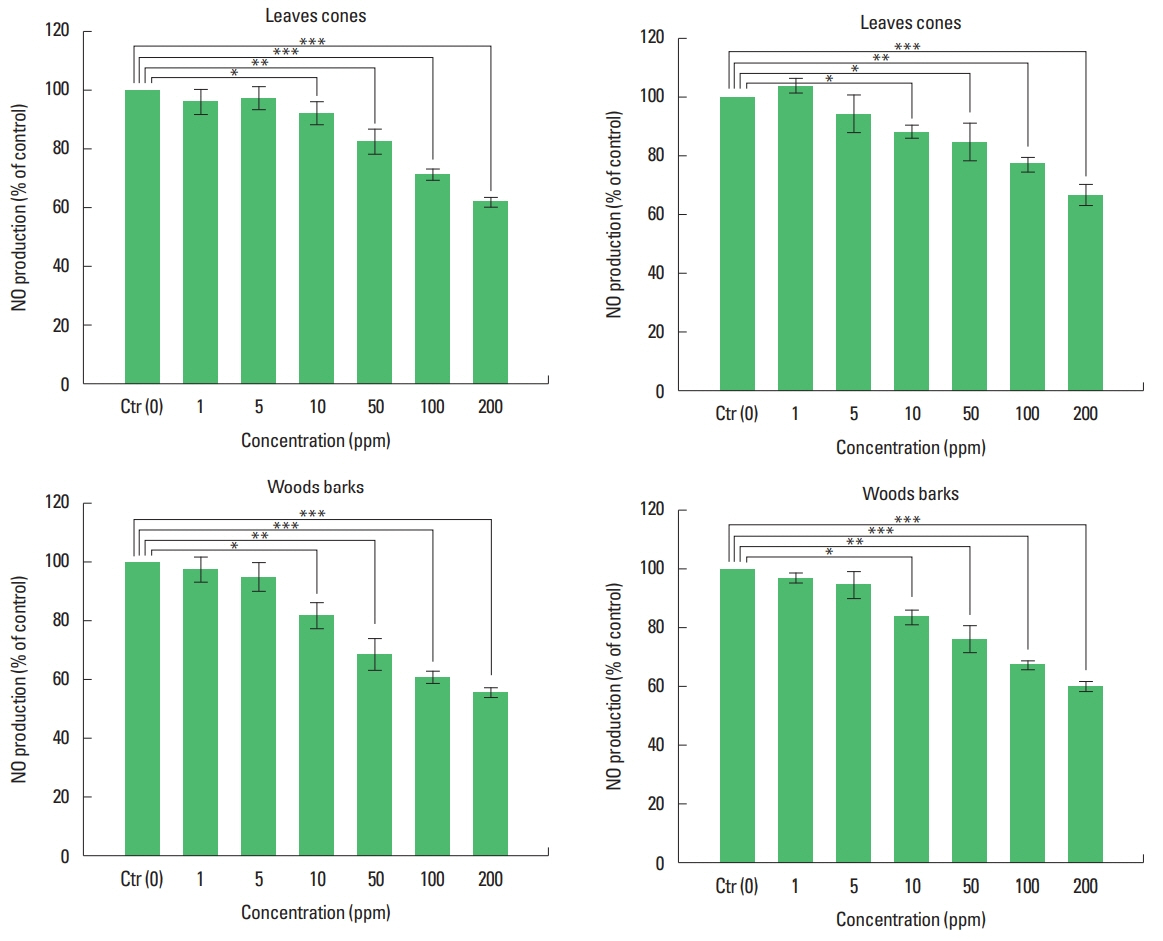
Fig. 6Inhibition of nitric oxide (NO) production on RAW 264.7 cells by α-pinene. Nitric oxide production was activated by lipopolysaccharide in RAW 264.7 cells. **P<0.01, ***P<0.001 vs. control. The data were obtained from 3 independent experiments. 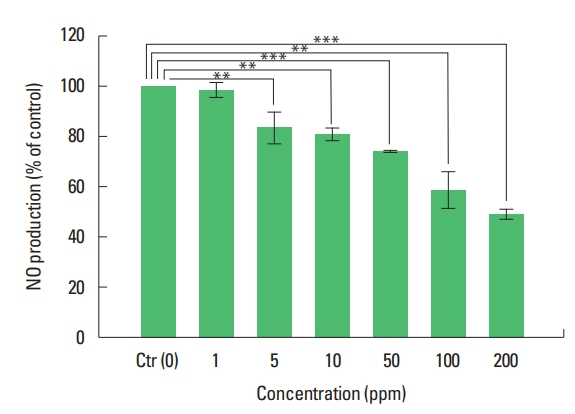
Table 1Fatty acid composition of each part of extracts from Chamaecyparis obtusa
Table 2Chemical composition of the extract of Chamaecyparis obtusa by supercritical fluid extraction |
|
|||||||||||||||||||||||||||||||||||||||||||