INTRODUCTION
Atopic dermatitis (AD) is a common chronic or relapsing inflammatory disease associated with pruritic and eczematous skin lesions (Boguniewicz and Leung, 2010; Leung et al., 2004; Savinko et al., 2012). In recent years, the occurrence of AD has rapidly increased in developing and developed countries (Odhiambo et al., 2009; Williams et al., 2008). The etiology of AD is not clear. However, it is thought to be linked with multifaceted interactions between genetic and environmental factors such as allergens and microbes, as well as between immune and skin barrier dysfunction.
It has also been reported that acute phase AD is strictly related to T helper 2 (Th2) cell-mediated lesions, and that chronic AD is related to Th1 cell-mediated AD lesions (Nilsson et al., 2011; Savinko et al., 2012). Furthermore, stimulated mast cells, increased IgE, IgG, and pro-inflammatory cytokines play key roles in the formation of skin lesions (Niebuhr and Werfel, 2010; Parisi et al., 2013). Several studies have reported that cytokines, including interleukin (IL)-4, IL-4, IL-13, IL-31, and both tumor necrosis factor-alpha (TNF-α) and interferon-gamma (IFN-γ) are key players in acute and chronic AD (Bilsborough et al., 2006; Vestergaard et al., 1999; Yamada et al., 1995). In addition, retinoic acid-related orphan receptor γT (RORγT) is an orphan nuclear receptor that controls the expression of Th17 cells. Consequently, IL-17 has been reported to play a role in certain skin conditions (Albanesi et al., 1999; Zheng et al., 2007). IL-17-producing Th17 cells are important for host immunity. Further, abnormal activation of Th17 cells is responsible for inflammation (McKenzie et al., 2006). Transforming growth factor-beta (TGF-β) is a multifunctional cytokine that is expressed in most tissues, including the skin. TGF-β is secreted by several cell types, such as T cells, macrophages, endothelial cells, and keratinocytes, and is known to be involved in different types of skin inflammatory disorders (Khaheshi et al., 2011; Letterio and Roberts, 1998).
Physical exercise is well-known to improve health and prevent disease (Haskell et al., 2007). Systematic physical exercise can help to reduce body weight and play a key role in the prevention of coronary artery disease, stroke, hypertension, and osteoporosis (Booth and Lees, 2007). However, exercise usually causes sweating, which can result in itching. Many patients with AD are thought to refrain from exercise because it increases itching. Previous studies have shown that exercise and sweating are major exacerbating factors for patients with AD (Stern et al., 1998; Williams et al., 2004). Nevertheless, regular activity may beneficial to patients with AD (Salzer et al., 1994). However, the connection between low-intensity exercise and AD has not yet been studied.
In this study, therefore, we examined the effect of low-intensity tower climbing resistance exercise (TCRE) on AD lesions using a BALB/c model. The effects were evaluated by assessing ear thickness, histopathological changes including mast cell count, AD-related pro-inflammatory cytokines in ear tissue, and serum immunoglobulin (Ig) levels, as well as the size and weight of draining lymph nodes (dLNs) and nondraining lymph nodes (ndLNs), along with expression of pro-inflammatory cytokines in CD4+ T cells from dLN and ndLNs.
MATERIALS AND METHODS
Materials
TRIzol reagent for RNA extraction was purchased from Invitrogen (Carlsbad, CA, USA). Primary antibodies and peroxidase-conjugated secondary antibodies were purchased from Santa Cruz Biotechnology Inc. (Santa Cruz, CA, USA). All other reagents were of the highest grade that was commercially available at the time of the study.
Animals
Eight-week-old, female BALB/c mice were purchased from Samtako (Samtako Bio Korea Co. Ltd., Osan, Korea) and housed under specific pathogen-free conditions. The animals were housed 5–10 per cage in a laminar air-flow room, maintained at a temperature of 22°C±2°C, with a relative humidity of 55%±5% throughout the study. All experiments were approved by the Institutional Animal Care and Use Committee of Konkuk University (KU14012).
Induction of AD lesions in the ear and TCRE protocol
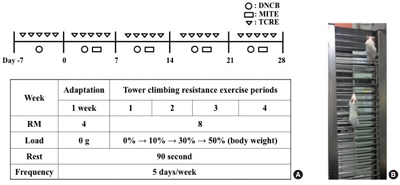
AD was induced in the mice by repeated local exposure of the ears to Dermatophagoides farinae extract (DFE; house dust mite extract) and 2,4-dinitrochlorobenzene (DNCB), as previously described (Kim et al., 2014). For the induction of AD, the mice were divided into four groups (control, TCRE, AD-only, AD+TCRE). The surfaces of both earlobes were stripped 5 times with surgical tape (Nichiban, Tokyo, Japan). After stripping, 20 μL of 1% DNCB was painted onto each ear, followed 4 days later by 20 μL of DFE (10 mg/mL). DFE or DNCB treatment was administered alternately once per week for 4 weeks.
The mice climbed the vertical ladder for 4 weeks. The exercise was accomplished utilizing a 1-m ladder with a 1.5-cm grid and inclined at 85°. Initially, the mice climbed with free weights for a week, to become accustomed. For the first training session, a free weight equivalent to 10% of their body weight was attached to the base of their tail, and the resistance was progressively increased to 30%, 50% for 4 weeks. When the mice reached the top of the ladder, they were allowed to rest for 90 sec. The training session was stopped when the mice succeeded to climb the ladder for eight repetitions.
Ear thickness measurement and blood sample preparation
Ear thickness was measured 24 hr after DNCB or DFE application with a dial thickness gauge (Kori Seiki MFG, Co., Tokyo, Japan). At days 14 and 28, blood samples were collected by the orbital puncture. Plasma samples were prepared from the blood samples and stored at −70°C for further analysis. After blood collection, the ears were removed and used for histopathological analysis. Serum IgE and IgG2a levels were measured at days 14 and 28 after the first induction, using an IgE enzyme-linked immunoassay kit (Bethyl Laboratories Inc., Montgomery, TX, USA) according to the manufacturer’s instructions.
Histological observations
Excised ears were fixed in 4% paraformaldehyde for 16 hr and embedded in paraffin. Thin (6 μm) sections were stained with hematoxylin and eosin (H&E). The thickness of the epidermis and dermis was measured under a microscope. For measurement of mast cell infiltration, skin sections were stained with toluidine blue, after which the number of mast cells was counted in five randomly chosen fields of view.
Real-time polymerase chain reaction
Quantitative real-time polymerase chain reaction (PCR) was carried out using a Thermal Cycler Dice TP850 (Takara Bio Inc., Shiga, Japan) according to the manufacturer’s protocol. Total RNA was isolated from the ear tissue and lymph nodes of each group. The PCR conditions were similar to those previously described (Kim et al., 2014). Briefly, 2 μL of cDNA (100 ng), 1 μL of sense and antisense primer solution (0.4 μM), 12.5 μL of SYBR Premix Ex Taq (Takara Bio Inc.), and 9.5 μL of dH2O were mixed to obtain a final 25-μL reaction mixture in each reaction tube. The amplification conditions were as follows: 10 sec at 95°C, 40 cycles of 5 sec at 95°C and 30 sec at 60°C, 15 sec at 95°C, 30 sec at 60°C, and 15 sec at 95°C. In each sample, the expression level of the analyzed gene was normalized to that of GAPDH and presented as a relative mRNA level.
Statistical analysis
Statistical analysis was carried out using SAS ver. 9.2 (SAS Institute, Cary, NC, USA). Multiple-group data were analyzed by one-way analysis of variance followed by Dunnett multiple range test. All results are expressed as the mean±standard deviation of comparative fold differences. Data are representative of three independent experiments. The threshold for significance was set at P<0.05.
RESULTS
Effect of TCRE on the ear thickness and histopathological observation and on serum Ig levels
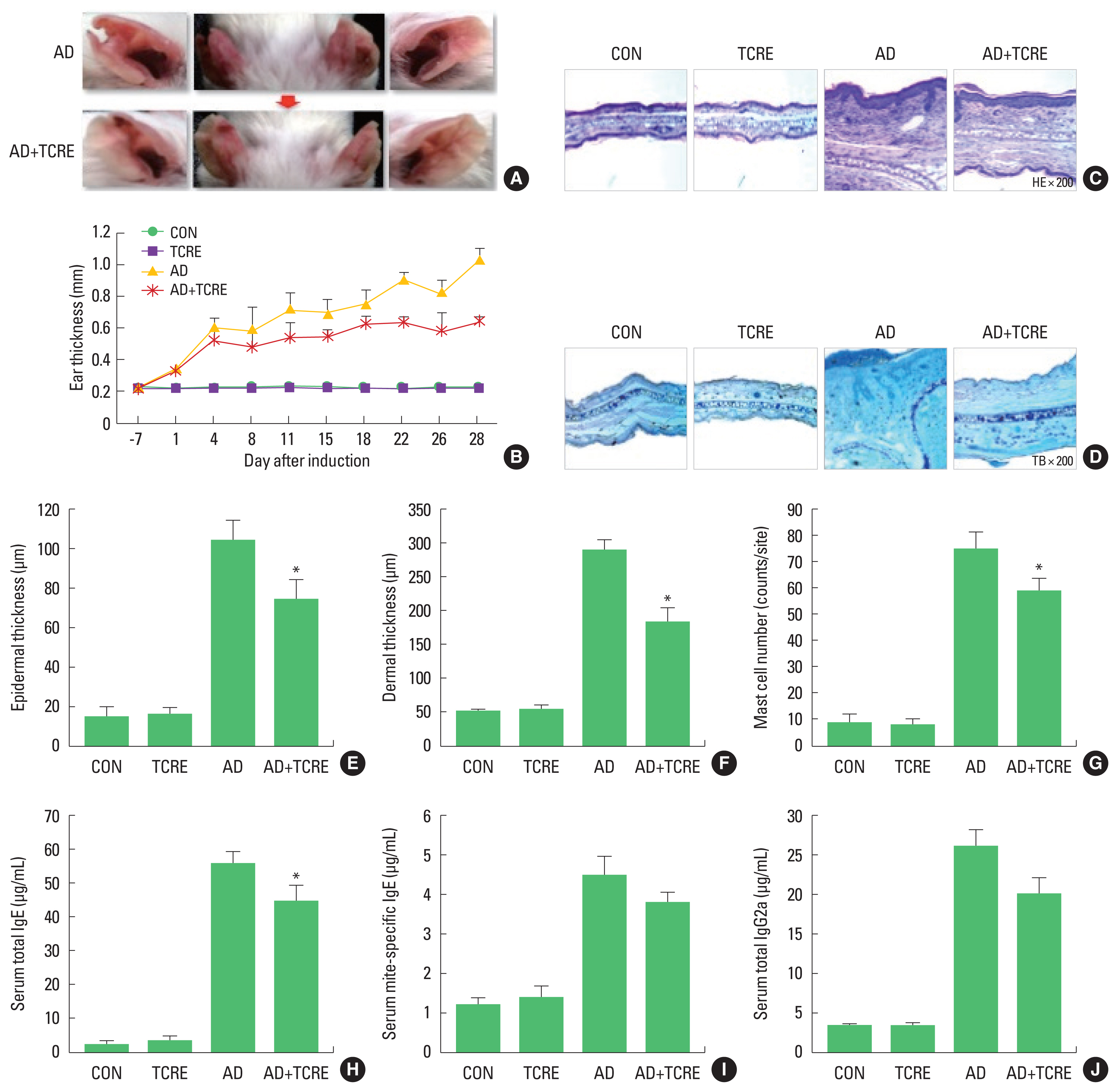
Experimental AD was induced in both earlobes of BALB/c mice by the alternative painting of DFE or DNCB for 4 weeks (Fig. 1A). To evaluate the immunomodulatory effects of TCRE, the mice climbed a vertical ladder for 4 weeks. For the first training session, a free weight equivalent of 10% of their body weight was attached to the base of the tail, and the resistance was progressively increased to 30%, 50% over 4 weeks (Fig. 1B). We found that DFE/DNCB application significantly increased ear thickness and AD lesions. On the contrary, following TCRE for 4 weeks, we observed that TCRE significantly reduced ear thickness (Fig. 2A, B). The TCRE group mice showed significantly reduced epidermal and dermal thickness (Fig. 2C, E, F). In addition, TCRE reduced numbers of infiltrating immune cells, such as mast cells, compared with the AD group (Fig. 2D, G).
Mice in the TCRE group showed significantly reduced total and specific IgE (Fig. 2H, I) and IgG2a levels (Fig. 2J) in comparison with those of DFE/DNCB-treated mice. These data suggest that the effect of TCRE in AD progression is related to downregulation of serum Ig levels.
Effect of TCRE on the expression of various pro-inflammatory cytokines in vivo
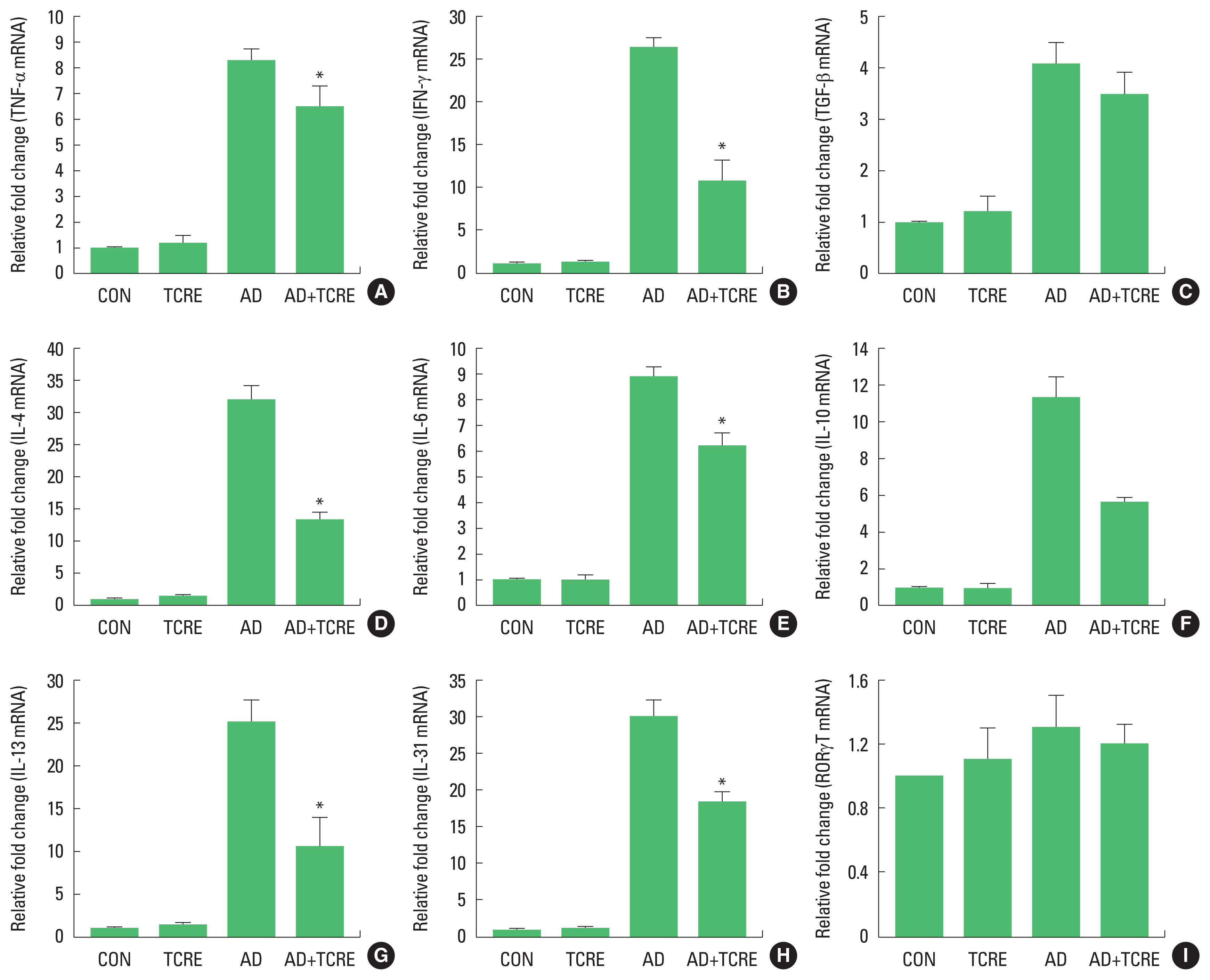
We further examined mRNA expression levels of AD-related pro-inflammatory cytokines from ear tissues by real-time PCR. As shown in Fig. 3, mRNA levels of all tested cytokines were upregulated in the ear tissue of AD mice. On the contrary, the expression of TNF-α, INF-γ, TGF-β1, and Th2-related cytokines such as IL-4, IL-6, IL-10, IL-13, and IL-31 were significantly suppressed in the AD+TCRE in the ear tissue. In addition, TCRE significantly reduced the expression of RORγT mRNA in the ear tissue (Fig. 3).
Accordingly, we examined the size and weight of dLNs and ndLNs as well as cytokine-related mRNA expression in CD4+ T cells from dLNs and ndLNs. We observed that AD mice had larger and heavier dLNs than healthy untreated control mice; after 4 weeks of TCRE, the size and weight of dLNs were reduced (Fig. 4A), and no significant differences were observed in the weight of ndLNs between mice in the TCRE and AD groups (Fig. 4A). Analysis of mRNA expression of pro-inflammatory cytokines by CD4+ T cells purified from dLNs showed that mice in the AD+TCRE group showed significantly lower dLN expression of TNF-α, IFN-γ, IL-4, IL-13, and IL-17 (Fig. 4B), whereas mRNA expression of pro-inflammatory cytokines was not changed in CD4+ T cells purified from ndLNs from the AD+TCRE group (Fig. 4C).
DISCUSSION
The benefits of exercise have been well studied. Regular physical exercise reduces the risk of several diseases and improves antioxidant activity and immune function (Booth and Lees, 2007; Sen, 1999). However, patients with AD may show a reduced desire for physical exercise due to itching following intense exercise. Therefore, in the present study, we assessed the influence of low-intensity exercise in an AD model.
The allergic response is linked to mast cells, which originate from myeloid stem cells. Therefore, we performed histological analysis of atopic ears to confirm the visual evaluation of AD signs. Ears were removed from each group of mice, then stained with H&E or toluidine blue and infiltration of mast cells, and thickening of the epidermis and dermis were observed under a microscope. After TCRE for 4 weeks, TCRE significantly decreased ear thickness epidermal and dermal thickness. In addition, TCRE also reduced numbers of infiltrating mast cells.
Meanwhile, most patients with AD have increased total serum immunoglobulin IgE levels and specific IgE antibodies to environmental allergens (Dokmeci and Herrick, 2008; Schäfer et al., 1999). Mast cells are stimulated by cross-linking of IgE and an allergen, which stimulates release of chemical mediators including cytokines at the sites of allergic reactions (Borish and Joseph, 1992). Further, high levels of IgG antibodies are associated with chronic AD, and IgG is related to the Th1 response (Mathlouthi and Koenig, 1986). Several studies have also reported that AD is linked to both Th2 and Th1 cell-mediated lesions and causes increased total IgE levels and Th2/Th1-type cytokine expression (Chen et al., 2004; Grewe et al., 1998; Hamid et al., 1994; Neis et al., 2006). To determine whether these effects are mainly exerted via the Th1 or Th2 response, serum levels of IgE (total and DFE-specific) and IgG2a were measured from each treatment group. Total and specific IgE, and IgG2a levels of AD+TCRE group were significantly reduced compared with AD group. In addition, TCRE significantly reduced not only Th2 cytokines but also the Th1-related cytokine TNF-α, IFN-γ and the pro-inflammatory cytokine TGF-β1 which was found to be altered in several skin disorders (Khaheshi et al., 2011). Moreover, RORγT is essential for the induction of IL-17 transcription and is an indicator of Th17-dependent autoimmune disease in mice. Here, TCRE decreased the RORγT level.
Naive CD4+ T cells cause the generation of Th1 and Th2 cells in the presence of pathogens. Th1 cells produce TNF-α and IFN-γ, whereas Th2 cells secrete IL-4, IL-13, and IL-17 which promote humoral immunity and IgE production, as well as regulate Th1 response (Abbas et al., 1996). In particular, IL-4 plays a key role in transforming naive CD4+ T cells into Th2 cells (Rincón et al., 1997). Since AD often develops as a systemic disease (Darlenski et al., 2014), we next examined whether TCRE affected systemic immune responses. Larger dLNs were found in mice in the AD+TCRE group, compared with mice in the AD group, and no significant differences were observed in the weight of ndLNs between mice in the TCRE and AD groups. Further, mice in the AD+TCRE group showed reduced mRNA expression of TNF-α, IFN-γ, IL-4, IL-13, and IL-17.
Meanwhile, previous studies have reported that exercise and sweating are noteworthy exacerbating factors for the schoolchildren with AD (Stern et al., 1998; Williams et al., 2004). Tay et al. (2002) have also studied AD in Singaporean schoolchildren and reported that exercise, heat, and sweating are the most irritating factors for schoolchildren. Accordingly, Kim et al. (2014) showed that high-intensity swimming exercise increases AD symptoms in BALB/c mice by increasing Ig and cytokine expression. Contrary to these data, the results of our study show that low-intensity exercise decreases AD symptoms.
In this study, we showed that low-intensity exercise reduces the intensity of DFE/DNCB-induced AD-like skin lesions in BALB/c mice. TCRE downregulated the severity of histopathological symptoms, production of Ig, and mRNA expression of pro-inflammatory cytokines in ear tissue, and reduced the size and weight of dLNs, along with expression of pro-inflammatory cytokines in CD4+ T cells from dLNs. Taken together, our data suggest that TCRE may be beneficial for patients with AD.